Series of videos that can be used in a Chemistry class created by Paul Anderson- Bozeman Science
- Subject:
- Chemistry
- Physical Science
- Material Type:
- Lecture
- Provider:
- Bozeman Science
- Date Added:
- 11/23/2016

Series of videos that can be used in a Chemistry class created by Paul Anderson- Bozeman Science
This channel will feature chemistry lectures, problem-solving videos, and laboratory tips and tricks.
CHEM221 General Chemistry I
The first course in the general chemistry sequence for science, engineering and health pre-professional students. Topics include: measurement, atomic structure, molecular structure, chemical reactions, stoichiometry, and thermochemistry. This course includes a laboratory component.
Chemistry is all around us "from the air we breathe to the food we eat" to the items at the supermarket that say “no chemicals added”. In fact, it is impossible to create something without using chemistry because chemistry consists of all matter. It allows us to answer questions as simple as why a candle goes out when a glass is placed over it to more complex questions such as does a candle actually burn in zero gravity? Chemistry is the study of matter and the changes it undergoes such as ice changing from the solid to liquid to gas phase. People have used it for things such as creating metal from an ore, dying fabric and making cheese. Chemistry deals with different substances and how they can interact with each other to create a product.
As you begin your study of college chemistry, those of you who do not intend to become professional chemists may well wonder why you need to study chemistry. You will soon discover that a basic understanding of chemistry is useful in a wide range of disciplines and career paths. You will also discover that an understanding of chemistry helps you make informed decisions about many issues that affect you, your community, and your world. A major goal of this text is to demonstrate the importance of chemistry in your daily life and in our collective understanding of both the physical world we occupy and the biological realm of which we are apart.

A Chemistry Perspective for discussion of Environmental Issues
Short Description:
Chemistry and the Environment is designed as a resource to accompany lectures for an Environmental Studies course that explores current environmental issues from a chemical perspective. It was edited from the OpenStax book Chemistry 2e.
Long Description:
Chemistry and the Environment is designed as a resource to accompany lectures for an Environmental Studies course that explores current environmental issues from a chemical perspective. It was edited from the OpenStax book Chemistry 2e.
Chemistry 2e is designed to meet the scope and sequence requirements of the two-semester general chemistry course. The textbook provides an important opportunity for students to learn the core concepts of chemistry and understand how those concepts apply to their lives and the world around them. The book also includes a number of innovative features, including interactive exercises and real-world applications, designed to enhance student learning. The second edition has been revised to incorporate clearer, more current, and more dynamic explanations, while maintaining the same organization as the first edition. Substantial improvements have been made in the figures, illustrations, and example exercises that support the text narrative. Changes made in Chemistry 2e are described in the preface to help instructors transition to the second edition. The first edition of Chemistry by OpenStax is available in web view here.
Word Count: 166210
ISBN: 978-1-7777612-7-1
(Note: This resource's metadata has been created automatically by reformatting and/or combining the information that the author initially provided as part of a bulk import process.)

Chemistry and the Environment is designed to accompany a one-semester course in chemistry-based discussions of important environmental issues such as air pollution, the ozone layer, climate change and water quality. Chemical principles are introduced, followed by environmental ‘focus’ sections to base discussions on the scientific principles and societal intricacies of the individual topics. Instructors can also use the focus sections as a resource for presentation slides. Chemistry 2e is designed to meet the scope and sequence requirements of the two-semester general chemistry course. The textbook provides an important opportunity for students to learn the core concepts of chemistry and understand how those concepts apply to their lives and the world around them.
People around the world are fascinated about the preparation of food for eating. There are countless cooking books, TV shows, celebrity chefs and kitchen gadgets that make cooking an enjoyable activity for everyone. The chemistry of cooking course seeks to understand the science behind our most popular meals by studying the behavior of atoms and molecules present in food. This book is intended to give students a basic understanding of the chemistry involved in cooking such as caramelization, Maillard reaction, acid-base reactions, catalysis, and fermentation. Students will be able to use chemistry language to describe the process of cooking, apply chemistry knowledge to solve questions related to food, and ultimately create their own recipes.

GS105B
Short Description:
This textbook presents introductory chemistry within the framework of food and cooking for a one-term general education course for non-science majors.
Long Description:
This textbook presents introductory chemistry within the framework of food and cooking for a one-term general education course for non-science majors.
Word Count: 47436
(Note: This resource's metadata has been created automatically by reformatting and/or combining the information that the author initially provided as part of a bulk import process.)

The seminar is designed to look at the science of triathlons and sports from a molecular/chemical biological point of view. We will be able to use our own bodies to see how exercise affects the system, through observations written in a training journal. We will also improve the overall fitness of the class through maintaining a physical fitness program over the course of the term. The end of the term will have us all participate in a mini-triathlon.

Matching game, including matching common ionic charges and ions and the compounds they form and naming of greek prefixes for covalent molecules.

This activity provides a demonstration and lab exploration of one of the main "building blocks" of the periodic table of elements: chlorine. During the lab, students compare physical and chemical properties of chlorine compounds.

This lesson has students solving the mystery of a note by conducting chromatography. They will test different washable markers and see that different marker colors are made up of various pigments. They will compare and contrast the note chromatography with other chromatography results from a variety of markers.

This is a laboratory investigation where students will use the scientific method to solve a new, experimental question in chromatography.
Students will be able to differentiate between pure substances and mixtures, as well as elements, compounds, homogeneous mixtures, and heterogeneous mixtures.

This infograph gives you the detailed information on classification of chromatographic methods. Chromatographic methods had 3 main types that is based on stationary, stationary phase format & mobile phase.
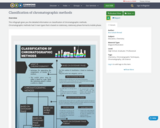
This infograph gives you the detailed information on classification of chromatographic methods. Chromatographic methods had 3 main types that is based on stationary, stationary phase format & mobile phase.
In this lesson, students further their knowledge of redox titrations while examining the pressures that contamination and climate change put on access to clean water.
Step 1 - Inquire: Students observe a field scientist testing the dissolved oxygen content of the Hudson River and generate questions.
Step 2 - Investigate: Students apply the Winkler Method as a tool for assessing the health of bodies of water and identify the stresses placed on water sources by climate change.
Step 3 - Inspire: Students explore Sustainable Development Goal #6 and consider what steps they can take to protect the drinking water in their communities.

This article continues an examination of each of the seven essential principles of climate literacy on which the online magazine Beyond Weather and the Water Cycle is structured. Principle 2 covers the complex interactions among the components of the Earth system. The author discusses the scientific concepts underlying the interactions and expands the discussion with diagrams, photos, and online resources.

This question is addressed through a series of questions and answers, each providing related introductory information such as how climate change is studied, the history of Earth’s climate, and the effects of climate change on Earth’s geology and biology. The Climate Kids website is a NASA education resource featuring articles, videos, images and games focused on the science of climate change.

Use the imbedded sliding scale to observe changes in sea ice distribution, carbon emission levels and average global temperatures over time, as well as the effects of variations in sea level rise along coastal regions. This interactive is part of the Climate Kids website, a NASA education resource featuring articles, videos, images and games focused on the science of climate change.
This short video discusses where carbon dioxide, the gas that is mainly responsible for warming up our planet and changing the climate, comes from. It discusses how the rise in atmospheric carbon dioxide comes directly from the burning of fossil fuels and indirectly from the human need for energy.