- Author:
- Amy Petros
- Subject:
- Chemistry
- Material Type:
- Module
- Level:
- Community College / Lower Division, College / Upper Division
- Tags:
- License:
- Creative Commons Attribution
- Language:
- English
- Media Formats:
- Text/HTML, Video
Mini-lecture: Drawing Lewis structures using Connect the Dots
Mini-lecture: Exceptions to the Octet Rule
Mini-lecture: How to assign formal charge
Mini-lecture: How to draw Lewis structures for carbon dioxide, carbon monoxide, and ozone
Mini-lecture: how to identify valence electrons
Mini-Lecture: Lewis structures with multiple bonds (oxygen and nitrogen)
Ted Ed: How Atoms Bond
Lewis Structures
Overview
This multi-part module introduces covalent bonding and Lewis structures as a model of covalent bonding.
Starting with valence electrons, a method of connecting unpaired electrons and/or redistributing valence electrons to satisfy the octet rule is introduced.
Numerous examples are presented including CO, ozone, and polyatomic ions
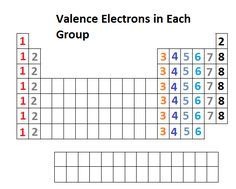
Identifying valence electrons and introduction to bonding
Before we can start drawing Lewis structures, we need to identify how many valence electrons (the ones typically involved in covalent bonding) each element has.
The mini-lecture includes two tools to identify valence electrons. The Ted Ed video provides an introduction to bonding.
How to Draw a Lewis Structure
This set of mini-lectures shows an alternative method to drawing Lewis structures rather than the traditional: add up all valence electrons, attach all atoms with a single bond and subtract 2 electrons per bond, etc.
This mini-lecture shows a method for drawing Lewis structures that is different from the typical approach of summing all the valence electrons and distributing out 2 at a time as bonds and lone pairs.
Additional mini-lectures are attached, which show more examples including exceptions to the octet rule, polyatomic ions and atypical bonding examples (ie: carbon monoxide).
How to Assign Formal Charge
In the video, we eschew the typical format of the Formal Charge equation and simplify into:
FC = # valence electrons - # lines - # dots
lines = covalent bond in the Lewis structure
dots = lone pair (individual) electrons
Students get confused based on the various ways this formula is written, so we simplify to a visual model.
Now that we have a basic method for drawing Lewis structures, how to we pick the most accurate one if we can draw multiple structures that follow our rules?
Assigning Formal Charge helps us to identify the most reasonable Lewis structure.