- Author:
- SATHESHKUMAR KESAVAN
- Subject:
- Biology
- Material Type:
- Activity/Lab, Lecture Notes, Student Guide
- Level:
- Graduate / Professional
- Tags:
- License:
- cc-mark
- Language:
- English
collagen triple helix
Overview
The cellular microenvironment, characterized by an extracellular matrix (ECM), played an essential role in the transition from unicellularity to multicellularity in animals (metazoans), and in the subsequent evolution of diverse animal tissues and organs.
collagen
COLLAGEN
The extracellular matrix (ECM) played an essential role during the transition from unicellular organisms to multicellular animals (metazoans). The ECM comprises a basement membrane (BM) that underlies epithelia cells, and an interstitial matrix (IM) that is positioned between cells in the intercellular spaces and undergoes continuous controlled remodeling (Hynes, 2012; Bonnans et al., 2014; Nelson and Bissell, 2006; Inman et al., 2015). Yet, a major gap in cell biology is to understand how cells generate and interact with the ECM (Sherwood, 2015; Jayadev and Sherwood, 2017).
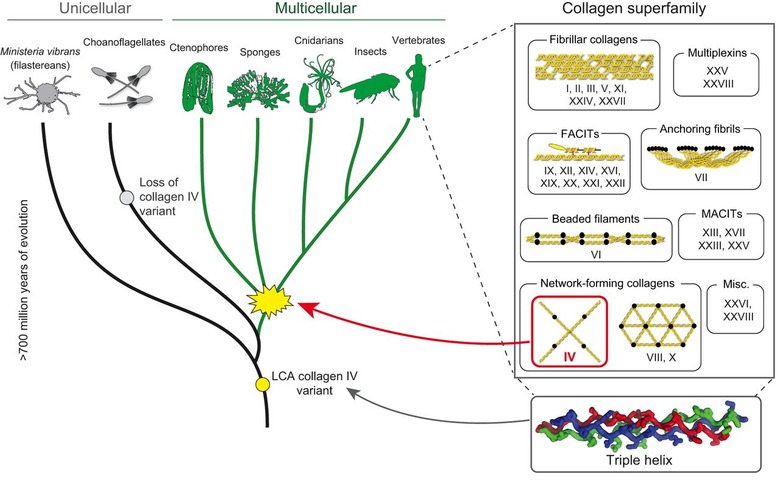
The collagen superfamily of proteins is a major component of ECMs, which – in vertebrates – comprises 28 types (I–XXVIII) that are derived from a total of 46 α-chains across the superfamily (Fig. 1) (Ricard-Blum, 2011; Kadler et al., 2007; Ricard-Blum and Ruggiero, 2005). Invertebrates generally contain collagen IV, XV or XVIII, some fibrillar collagens, as well as some fibril-associated collagens with interrupted triple helices (FACITs) (Fidler et al., 2014, 2017; Fahey and Degnan, 2010; Meyer and Moussian, 2009; Boot-Handford and Tuckwell, 2003; Whittaker et al., 2006; Kadler et al., 2007). Among these collagens, type IV is the evolutionarily most ancient, based on recent studies of non-bilaterian animals (sponges, ctenophores, placozoans and cnidarians) and unicellular groups (Fidler et al., 2017; Grau-Bove et al., 2017)
Collagens are the most abundant protein in the human body (Kadler et al., 2007; Shoulders and Raines, 2009). They occur as diverse supramolecular assemblies, ranging from networks to fibrils, and broadly function in structural, mechanical and organizational roles that define tissue architecture and influence cellular behavior (Shoulders and Raines, 2009; Ricard-Blum, 2011; Ricard-Blum and Ruggiero, 2005). Defects in collagens underlie the cause of almost 40 human genetic diseases, affecting numerous organs and tissues in millions of people worldwide.