- Author:
- Brandy Hughes
- Subject:
- Chemistry
- Material Type:
- Activity/Lab, Interactive, Reading
- Level:
- High School
- Tags:
- License:
- Creative Commons Attribution
- Language:
- English
- Media Formats:
- Interactive, Text/HTML, Video
Balancing Chemical Equations
Changing Matter Not Weight
Chapter 7 Notes
Law of Conservation of Mass Lab
Mathematically Balancing Equations
Law of Conservation of Mass
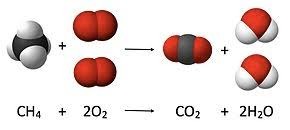
Overview
This unit dicusses the law of conservation of mass, as well as teaches the fundamentals of balancing chemical equations.
Gum Lab
Students determine the portion of original mass of gum that is swallowed for sugar and sugar-free gum.
Changing Matter Not Weight
Matter is not created nor destroyed; it simply changes from one form to another. This law of conservation of mass challenges elementary students’ ideas about matter, because many children may think that matter is created or destroyed in a chemical reaction. In this lesson, students will challenge their preconceptions about matter by experimenting with physical and chemical changes to determine that the total weight of the matter does not change. Students will use math to show that the total weight of matter is equal to the sum of the weight of its component parts, and they will graph this information to show that the weight of matter is conserved during physical and chemical changes.
Notes PowerPoint
Follow the Powerpoint, and take notes. Make sure to include all vocabulary.
Balancing Chemical Equations Article
This lesson will reinforce your knowledge about chemical formulas and introduce the concept of balanced chemical equations.
Balancing Chemical Equations Activity
How do you know if a chemical equation is balanced? What can you change to balance an equation? Play a game to test your ideas!
Video
Mathematically Balancing Equations
This provides a mathematical method of balancing chemical equations as an alternative to the conventional method (by inspection)