- Author:
- Amrita Madabushi
- Subject:
- Biology
- Material Type:
- Activity/Lab, Lecture Notes, Module
- Level:
- Community College / Lower Division
- Tags:
- License:
- Creative Commons Attribution
- Language:
- English
Biotech Lab Techniques Manual

Overview
This is a laboratory manual for Biotechnology lab at the undergraduate level.
Welcome
Welcome to your first laboratory course in biotechnology Techniques of Instrumentation for Biotechnology (BTC-105). The course is designed for you to understand, learn and practice techniques and instruments commonly used in biotechnology. You will also learn and master labmath required to make buffers, media and dilutions related to molarity, percentage and other concentrations units.
The objectives of the course are:
- Understand the concepts underlying techniques & instrumentation in biotechnology.
- Apply formulas to calculate, prepare and standardize various strengths of molar, normal, and percent solutions within a 10% error.
- Operate instruments in the average biological, chemical, clinical, and/or analytical laboratory.
- Master in Scientific presentations and writing generating lab reports using good documentation practices.
- Learn to work efficiently independently as well as in teams.
- Develop critical thinking and analytical abilities to perform small and big projects.
Students are expected to behave professionally in a laboratory at all times. The lecture will provide background and relevant information about the solutions, prep, procedure, and related techniques. The students should plan on having the following in the lab at all times- a lab coat, close-toed shoes, and a bound lab notebook.
The techniques you will learn in this course include the following
- Making Buffers
- Simple and serial Dilutions
- pH meter
- Thin layer Chromatography.
- Spectrophotometer
- Centrifuge
- Protein estimation by Bradford.
- Gel filtration chromatography
- Bacterial Cell – Growth Curve, Transformation
- Mammalian cell culture
- Polymerase chain reaction
- DNA gel
- Protein gel
Please make sure to ask questions if you are unaware of a certain technique or where a chemical is in teh lab.
Introduction to Laboratory
General Lab Organization & Procedures
This module will serve as an introduction to the lab. There are different kinds of labs based on their research and work focus. The type of labs are:
- Research Labs (University) – focus on a question or hypothesis related to basic science or applied research.
- Applied Science Labs (Company)- to develop & make products for human diseases, drug discovery, also including biologically engineered food and medical products.
- Core Labs –supports the research of others using a specific type of assay or in an area of expertise.
- Clinical Labs - the majority of work in these labs supports patient care.
Figure 1: Different types of scientific labs based on the research work.
Laboratory Personnel work in a laboratory at different level as shown in the figure below. These levels are primarily based on the education level of individuals. So as an Associate or certificate student in Biotechnology, you will fall in the same category as undergraduate student researcher. You can find employment as lab technician.
Lab Routine:
It is important to understand the lab routine for any lab. In most of the labs you are expected to know what is the dress code and what personal protective equipment (PPE) you are supposed to wear. In this lab you must wear a lab coat on top of your clothes and also closed toed shoes. Every lab is different, so make sure to
Before entering the lab, please ensure:
- Leave the food/ drinks outside the lab.
- Have your lab coat and lab notebook with you.
While in the lab:
- Wear your lab coat and take it off if you have to step out of the lab even for a few minutes.
- Note down the name of instruments and ask if you have any doubts or questions about their usage.
- Note down the readings and observations of your experiments carefully.
Before leaving the lab:
- Make sure that you leave your bench clean.
- Use Lysol and scrub at the end of the lab work to wipe your working area.
- Make sure you keep all the unused lab items back on the shelves.
- Ensure that the used/dirty dishes are on the cart next to -20. (after rinsing and removing labels)
- Paper and other regular trash should be in the trash can.
- Chemical, media, and bacterial work-generated trash must be disposed in biohazard bags.
- Make sure you wash your hands before you go.
Find about the following item locations and practices before you start working:
- Chemicals
- First Aid box
- Glassware
- Fire extinguisher
- Lab coats
- Gloves
- Trash Disposal
- Computer usage
LABORATORY Dos and Don'ts
DO's:
- Take notes
- Wear lab coat and PPE as needed.
- Familiarize yourself with lab
- Conduct experiments carefully.
- Different trash in different disposal bins.
- Cleanup after you complete experiments.
Don'ts:
- Use cell phones.
- Use reagents/ buffers without asking.
- Lab coats and gloves outside the lab if directed.
- No open toed shoes, shorts.
Methods to reduce the risk in laboratory work:
- NO eating
- NO smoking
- NO drinking
- NO chewing gum
- NO applying makeup
- NO combing hair.
Universal precautions in a laboratory:
Universal precautions have been developed to protect health professionals
- Most often applied in a clinical setting
- These are also important (outside the lab) for field epidemiology practices during an outbreak investigation (e.g., collecting lab specimens)
- Include hand hygiene, gloves, gown, masks, eye protection, face shields, safe injection practices
- Require that all equipment or contaminated items are handled to prevent transmission of infectious agents
- Special circumstances may require additional precautions
- For example, protective clothing, special site decontamination
Barriers
There are two types of barriers in a laboratory - Primary (physical) and Secondary (structural)
- Primary barriers are physical barriers or personal protective equipment (PPE) between lab workers and pathogens.
- Personal Protective Equipment or "PPE" is equipment worn by lab personnel to minimize exposure to hazards in the lab. Contact with chemical, radiological, physical, electrical, mechanical, or other workplace hazards can cause injury or illness. Personal protective equipment is worn according to biosafety levels and may include gloves, safety glasses and shoes, or full body suits.
- Secondary barriers are structural aspects of the laboratory that make the working environment safer against infection. For example, sinks for handwashing, special containment areas, special air ventilation patterns
These barriers will vary based on the microbes (biological agents) and chemicals being handled in the laboratory.
Biosafety Levels (BSL)
Biosafety level is a level of the biocontainment precautions required to isolate dangerous biological agents in an enclosed laboratory facility.
- BSL-1 agents are not known to cause disease in healthy adults (Example: B. subtilis, E. coli)
- BSL-2 agents are associated with human diseases derived from blood or body fluids (Example: Hepatitis B virus, C. botulinum)
- BSL-3 agents have the potential for respiratory transmission and may cause severe and potentially lethal infection (Example: M. tuberculosis, C. burnetti)
- BSL-4 agents are dangerous and exotic agents and have the potential to cause a high risk of life-threatening disease (Example: Ebola virus, Marburg virus)
Lab Safety – Signs & Symbols
Laboratory signs and symbols are vital for the safety of the lab and the lab personnel. Labs typically contain hazardous chemicals, biohazards, delicate equipment, and flammable objects. Having adequate signage helps lab personnel remind and prevent errors and accidents. The symbols are mostly self-explanatory. Some of the common lab symbols are shown below.
NFPA (National Fire Protection Association) Diamonds and HMIS Hazardous Material Identification System) Bars are Color & Number Coded with Hazard Information. NFPA signs alert first responders to chemical hazards.

DOT (Department of Transportation) Symbols Are Usually Found on Shipping Cartons.
MSDS: You must know information about a chemical prior to using it. Information is provided in Material Safety Datasheet (MSDS). You can look up MSDS of a chemical online. Some labs might have it stored in a physical file.
Exposure to Hazardous Chemicals can Result in acute or chronic health effects.
Acute – Effects occurring within hours or days of exposure
Chronic – Effects occurring after exposure over many years
Physical Hazards
- Fire
- Bunsen Burners
- Autoclaves
- Compressed Gas Cylinders
- Broken Glassware
- Razorblades and needles
- Electrical Equipment
- Ultraviolet light
Some terms used in the lab (do you know what these are?) :
- MSDS
- PPE
- Sharps
- BSL
- Biohazard
Laboratory Documentation
Contents of a Lab Notebook: Every lab has its documentation style to record experimental procedures and data. In this lab, you will record experiments as you do them weekly. For every lab, you are expected to record – Date, Aim, Materials, Procedure, Results, and Conclusions. Here is the purpose of each of them.
Aim – States the goal of the experiment.
Material – Includes all the chemicals and biological items that are required for the experiment
Procedure - Description/protocol with reference to calculations (such as molecular weight, concentrations, dilutions, etc.). All the experiment steps should be written carefully so someone can repeat that in the future and produce a similar outcome.
Observation – Includes the summary of data, often in form of a table, graph or figures.
Results -must include both qualitative and quantitative data. If the results are unexpected or negative, they should always be included.
Conclusions – Summary of the experimental results.
Pre-lab write-up is done before you begin the experiment (see below).
Lab notes which include the standard operating procedure (SOP) used, the data and detailed observations you make while doing the lab, and any other comments you may want to remember or convey to others
Post-lab Analysis involves any calculations, conclusions drawn, and questions answered after the lab is completed. Most lab exercises come with a set of analysis questions to be answered.
Good Documentation Practices
Documentation is the mechanism by which raw data are captured and evaluated to ensure personal accountability.
Good Documentation Practices or GDP provides a tangible, detailed record of observations and activities – a "paper trail." This is extremely important in a biotech or pharmaceutical company where products are being manufactured. The Food & Drug Administration (FDA) can supervise the process and products.
FDA assumptions about documentation must always be followed:
- If it isn't written down, it wasn't done
- If it isn't legible, you made a mistake
- If it is written in pencil, it was changed
- If it isn't "controlled," you made a copy.
- If it wasn't "approved" before you started, you made it up
Document dates in a standardized format must be specified in your Standard Operating Procedure.
Blank fields can be interpreted as missing data, and empty spaces at the bottom of a page (e.g. lab notebooks) can allow someone to add data without your knowledge or consent.
Good Documentation Practices in a lab notebook
We will try to follow the Good Documentation Practices in laboratory notebooks by trying to follow as many as items stated below:
- Use indelible (water-resistant) blue or black ink so that it doesn't fade over time or smudge if it gets wet. No pencils or felt-tipped markers are allowed.
- NEVER back date!
- Enter data directly in the controlled record and never on a piece of scrap paper in the interim.
- Make sure the entries are accurate and legible so that anyone can understand your entries.
- Draw a diagonal line through any blank fields or empty spaces at the bottom of a page and include 'N/A' along with your initials and date.
- Number each page in a controlled notebook chronologically. This verifies that no pages are missing.
- For instrument printouts and attachments make sure they are within the notebook limits, and are stuck with clear adhesive tape. Include your initials and date so that part of your initials will be on the attachment and part will be on the notebook.
- When entering repetitive data, do not use ditto marks.
Lab Math - Significant Figures, SI Unit/ Metric System
In this module, we are going to learn about more concepts that are relevant to laboratory math including significant figures, SI units, and metric systems including conversions within the metric system.
Lab math
Why is Lab Math Important?
PURPOSE
The purpose of this module is to become well versed with simple calculations
- Multiplication with factors of 10
- Division by factors of 10.
- How to move around decimal when multiplying or dividing.
- Remembering tables
- Multiplication by factors of 10
- When there is no decimal in a number then you can add the same number of zeros to the right as many you see in the multiplying factor of 10.
1. 1357 x 10 = 13570
2. 123 x 1000 = 123000
3. 8 x 200 ?
- When there is a decimal in a number then you can move the decimal to as many places to the right as the number of zeros in the multiplying factor of 10.
1. 135.7 X 10 =
2. 13.57 x 10 =
3. 1.357 x 1000 =
Division by factors of 10
- When there are zeros in number, equal number of zeros on top and bottom can cancel out.
200/10=
5000/1000=
- When there is no decimal in a number then you can assume that the decimal is in the end. Move the decimal to the left as many zeros as you see in the dividing factor of 10.
1357 /10 =
123 / 1000 =
8 / 200 ?
MULTIPLICATION PRACTICE
|
PRACTICE
1. 23 x 10 =
2. 12 x 100 =
3. 150 x 100 =
4. 230 x 10000 =
5. 12.3 x 100=
6. 1.567 x 10=
7. 180.5 x100 =
8. 3 x 300 =
9. 5 x 150 =
10. 300 x 500 =
11. 55/10 =
12. 330/100 =
13. 500/ 100 =
14. 660/1000=
15. 200/20=
16. 400/20 =
17. 10/10000=
18. 200/100000=
19. 120/12=
20. 103/100=
What kind of Lab Math will we be doing ?
- Scientific Notations
- Standard Notations
Standard & Scientific Notation
- Scientific numbers are written in 10x.
- The first number before x should be between 1 and 9.9999 (less than 10).
- Then count the numbers after that and put the count as a power of 10. Example : 2300 = 2.3 X 103
0.023 = 2.3 X 10-2
- Standard numbers are written in terms of 0s and decimal. Example : 4.5 X 103 = 4500
4.5 X 10-2 = 0.045
Symbol | Units | Standard Notation | Scientific Notation |
K | Kilo | 1000 | 1 x 103 |
100 | 1 x 102 | ||
10 | 1 x 101 | ||
m/g/l/M | 1 | 1 x 100 | |
0.1 | 1 x 10-1 | ||
c | Centi | 0.01 | 1 x 10-2 |
m | Mili | 0.001 | 1 x 10-3 |
0.0001 | 1 x 10-4 | ||
0.00001 | 1 x 10-5 | ||
m | Micro | 0.000001 | 1 x 10-6 |
0.0000001 | 1 x 10-7 | ||
0.00000001 | 1 x 10-8 | ||
n | Nano | 0.000000001 | 1 x 10-9 |
Scientific notation is a way of expressing really big numbers or really small numbers that are in standard notation.
- The scientific notation consists of two parts:
- A number between 1 and 10 (N)
- A power of 10 (x)
N x 10x
Example: Express in Scientific notation (a) 200,000 (b) 0.00002
Class Assignment
Brainstorming Practice Session
Scientific Figures & Scientific Notations
Write the appropriate scientific notation for the following
1. 600000 | = |
2. 0.0678 | = |
3. 23.785 | = |
4. 89600000 | = |
5. 0.000009 | = |
6. 50005 | = |
7. 543.00 | = |
8. 0.00456 | = |
Write the appropriate standard notation for the following
1. 3 x 10-3 =
2. 1.34 x 107 =
3. 7.29 x 10-2 =
4. 5.55 x 102 =
5. 1.054 x 101 =
6. 1.5 X 100 =
7. 2.05 x 105 =
8. 8.0354 x 10-4 =
Significant Figures
- There are 2 different types of numbers
- Exact
- Measured
- Exact numbers are infinitely important
- Measured number = they are measured with a measuring device (name all 4) so these numbers have ERROR.
- Solution
- Exact numbers are obtained by
- counting
- definition
B. Measured numbers are obtained by using a measuring tool
- Learning Check
Classify each of the following as an exact or a
measured number.(Hint: Whatever needs a measuring tool can not be exact number) 1 yard = 3 feet
The diameter of a red blood cell is 6 x 10-4 cm. There are 6 hats on the shelf.
Gold melts at 1064°C.
Measurement and Significant Figures
- Every experimental measurement has a degree of uncertainty.
- The best guess is needed for the tenth place.

What is the Length? | |
|
We can see the markings between 1.6-1.7cm
- We can't see the markings between the .6-.7
- We must guess between .6 & .7
- We record 1.67 cm as our measurement
- The last digit an 7 was our guess...
Measured Numbers
- Do you see why Measured Numbers have error…you have to make that Guess!
- All but one of the significant figures are known with certainty. The last significant figure is only the best possible estimate.
- To indicate the precision of a measurement, the value recorded should use all the digits known with certainty.
Below are two measurements of the mass of the same object. The same quantity is being described at two different levels of precision or certainty.

RULES: Counting Significant figures
- Non-Zero Digits: All the digits that are not zero count towards the number of significant figures.
Example 1: 324 | (3) Sig. Fig | |
2: 1.223 | (4) | |
3: 12.1256 | (6) | |
4: 1200001 | (7) |
- Zero Digits:
- Zeros that come between two non-zero numbers are counted in significant figures.
E.g. 10001 (5 Significant Fig)
- Zeros that come after non-zero number in decimal count. E.g. 2.10 (3 Significant Fig).
- Zeros that come before decimal do not count. E.g. 0.25 (2 Significant Fig.)
- Placeholder zeros do not count.
E.g. 0.00023 (2 significant figures)
1. Trailing zeros are significant only if the number has a decimal, otherwise not.
E.g. 1200.0 (5 significant Fig.); 1200 (2 Significant Fig.)
RULES: Significant figures after
Addition/ Subtraction/ Multiplication/ Division.
- When you multiply, divide, add or subtract 2 numbers having different significant figures then the final answer will have the minimum of the two significant figures:
E.g. 1.05 (significant figure 3) x 2.2 (significant fig 2) = 23.1 (Significant figure 2)
Rounding Off: After decimal number greater than 5 can be rounded to next digit.
E.g. 1.55 to 1.6 3.54678 to 3.5468
- Rounding Off: After decimal number lesser than 5 can be rounded to the previous digit.
E.g. 1.54 to 1.5 3.54674 to 3.5467
In the last module, we learned about the concept of significant figures. Significant figures (or significant digits) are the digits in a number of a measurement that we can say is accurate and precise with a good degree of confidence.
RULES or counting Significant figures
WHAT COUNTS?
Non-Zero Digits: All the digits that are not zero counts towards the number of significant figures:
Example 1: 324 (3) Sig figs
2: 1.223 (4)
3: 12.1256 (6)
4: 1200001 (7)
Zero Digits count under the following conditions:
- Zeros that come between two non-zero numbers are counted in significant figures. E.g. 10001 ( 5 Significant Fig)
- Zeros that come after non-zero numbers in the decimal count. 2.10 ( 3 Significant Fig).
WHAT DOESN’T COUNT? (When Zero doesn’t count)
- Zeros that come before decimal do not count. E.g. 0.25 ( 2 Significant Fig.)
- Placeholder zeros do not count. E.g. 0.00023 ( 2 significant figures)
- Trailing zeros are significant only if the number has a decimal, otherwise not. E.g. 1200 (__ significant Fig.); 1200.0 (___ Significant Fig.)
RULES: Significant figures after Addition/ Subtraction/ Multiplication/ Division.
Rounding Off: After decimal number greater than 5 can be rounded to the next digit. Example 1.55 to 1.6 or 3.54678 to 3.5468
Rounding Off: After decimal, a number lesser than 5 can be rounded to the previous digit. E.g. 1.54 to 1.5 or 3.54674 to 3.5467
When you add or subtract two numbers having different significant figures then the final answer will be rounded of to the least number of decimal places
E.g. 1.05 (2 numbers after decimal) + 2.2 (1 number after decimal) = 3.25 rounded off to 3.2 (1 number after decimal)
When you multiply or divide two numbers having different significant figures then the final answer will have the minimum of the two significant figures:
E.g. 1.05 (3 significant figures) x 2.2 (2 significant figs) = 2.31 rounded off to 2.3 (2 Significant figures)
Practice Session- Significant Figures
What is the number of significant figures in the following?
- 39.57 _______
- 132 _______
- 2.90 _______
- 123.456 _______
- 0.23 _______
- 11.02 _______
- 18.500 _______
- 1230 _______
- 123000 _______
- 0.05000 _______
Round off:
- 76.512 to 4 significant figures. ________
- 932.058 to 5 significant figures. ________
- 0.5879 to 3 significant figures. ________
- 35.733 to 2 significant figures ________
- 120.5879 to 4 significant figures. ________
- 135.700 to 3 significant figures ________
- 0.5879 to 3 significant figures. ________
- 35.745 to 3 significant figures. ________
- 100.010 to 4 significant figures. ________
- 100.090 to 4 significant figures. ________
Write the answer in the appropriate significant figures
- 6.28 x 5.004 =
- 782.2 ¸ 2.36 =
- 5.789 + 3.21 =
- 6.789 – 5.980 =
SI Units & Metric System
The SI system is the International System of Units is the form of the metric system used around the world. There are seven basic units in the SI system: the meter (m), the kilogram (kg), the second (s), the kelvin (K), the ampere (A), the mole (mol), and the candela (cd). Important SI units that we should know in the context of laboratory math are listed here.
| SI Unit | Symbol | |
| Length | Meter | m |
| Mass | Kilogram | Kg |
| Volume | Liter | L |
| Time | Second | s |
| Amount of substance | Mole | mol/ M |
Metric System (Conversions)
Note how the major units are at every third place.
Why is the conversion important?
- There may be two different units on both sides of the equation. So you may need to convert
- Sometimes you may need to convert it to a unit that you can measure using lab equipment like a graduated cylinder or pipette.
How to convert within the metric system?
Always draw the metric system line before starting conversions.
1 Kg = ______________ g
3 g = _______________ mg
60 mg = ______________ug
1.2 Kg= _______________ g = _____________________mg
3.625 g = ________________mg = __________________ug
10.03 mg = ________________ug
5000g = ______________ Kg
3500 mg = _______________ g
6000 ug = ______________mg
12 g= _______________ Kg
362.5 mg = ________________g = __________________Kg
10.03 ug = ____________mg= ____________g=_____________Kg
Practice Session
Write the correct abbreviation for each metric unit.
1) Kilogram _____ 4) Milliliter _____ 7) Kilometer _____
2) Meter _____ 5) Millimeter _____ 8) Centimeter _____
3) Gram _____ 6) Liter _____ 9) Milligram _____
Try these conversions.
10) 2 gm = _______ mg 11) 5 L = _______ mL 12) 16 km = _______________m
13) 1000 m = __________ Km 14) 2000 g = _______ kg 15) 2500 ml = _______ L
16) 48 mm = __________Km 17) 75 mL = _____ L 18) 65 g = _________Kg
19) 5.6 mM = ___________µM 20) 0.55 M = _________ mM 21) 6.3 m = _____ mm
22) 8.2 mm = ___________ m 23) 5.6 M = _______ mM 24) 120 mg = _________ g
Pipette aids and pipet
In the laboratory, you may have to measure and dispense liquid of different types and quantities. This can be easily achieved using a device called a pipette which can draw liquid in the slender plastic or glass tube attachment known as a pipet.
Pipettes can be of different types: Bulb pipette, pipette pump, or pipette aid.
Bulb Pipettes are made of natural rubber bulbs that can help suction up to a fixed volume of liquid. These kinds of pipettes are accurate and yet cheap. This type of pipette aid maybe like a dropper or more complex device with three buttons: The first displaces air from the bulb, the second is used to draw liquid into the pipette, and the third is used to expel the liquid.
Pipette Pump are hollow plastic devices with a pump attached to one end and a pipet attached to the other end. The pump can be maneuvered with a thumb to regulate the amount of fluid that can be taken in the pipet.
Pipette Aid are motorized devices for suction and dispensing of accurate amount of liquid.
Pipets are glass (or plastic) tubes used for serology (serological pipet) and chemistry (volumetric pipet).
Volumetric Pipet
- Used to deliver a single specific volume of liquid, usually 1-100 ml.
- On a volumetric pipette, the specifications indicate how much liquid will be transferred if the liquid is drawn up to the calibration line on the neck
- the temperature at which the calibration was made
- whether it is a TD (To deliver) or TC (To contain) pipette
- After it is emptied, the small amount of liquid which remains in the tip should not be blown out.
- Volumetric pipettes are usually marked TD hence the liquid is allowed to drain out and these are NOT blow-out pipettes
Serological Pipet
Serological pipette are used for precise transfer of liquid the laboratory. They are called so because they were traditionally used in the laboratory for transfer of blood or serum.
- Printed on the neck of the pipette are the specifications that indicate:
- the maximum volume of liquid that can be transferred
- the size of the divisions on the pipette
- the temperature at which calibrations were made
- Always check the marking for TD or TC. TD pipettes are not blown out, TC pipettes are blown out.
The key difference between volumetric and serological pipettes is that volumetric pipettes are calibrated to deliver a certain volume of a solution (through free drainage), whereas serological pipettes are generally calibrated all the way to the tip, (and the last drop of the solution has to be blown out).
HANDLING STERILE PIPETTES
- When using sterile pipettes, be sure to use proper techniques.
- If you have a sterile package of disposable pipettes, tear only a small corner of the package open and push one pipette out of this opening, then immediately close the package to prevent contamination.
- If its taken from a steel container in a hood, keep the container closed after use immediately
Transferring a precise volume of liquid using a pipette
- A pipette bulb is used to draw liquid up into the pipet.
- There are many types of pipette bulbs.
- The use of 3 types will be demonstrated in this module. The first to be demonstrated will be the common rubber bulb.
Using the common rubber bulb
- Squeeze bulb and touch it to the mouth of the pipette.
- Place other end of the pipette in liquid to be transferred and slowly release pressure on bulb.
- Draw liquid up past desired level, quickly replacing bulb with index finger.
- Let liquid drain until bottom of meniscus lines up with desired level on pipette.
- Touch tip of pipette to inside of beaker to remove any adhering drops.
- Transfer liquid to second beaker and touch tip to inside of beaker and let liquid drain out of pipette.
Use of safety bulb pipette filler
Aspirate (A) → Suction (S) → Expel (E)
1: With a gentle twisting motion, insert the pipette into the Safety Pipette Filler about 1/2 cm.
2: To produce a vacuum for aspiration, squeeze valve “A” with your thumb and index finger of one hand while using your other hand to squeeze the bulb. “A” stands for “air” or “aspirate.”
3. To “pull” the liquid up into the pipette place the pipette into the liquid and squeeze the “S” or “suction” valve until the liquid reaches the desired level.
4. To “expel” the liquid, squeeze the “ E” valve. This will allow the liquid to flow out of the pipette except for the last drop.
Use of pipette pump and pipette aid
1. The first step is to connect a sterile pipet to the end.
2. The pipet mouth is then placed inside the liquid to be transferred. The mouth should never be inserted too much.
3. In case of the pipette pump, the upper part can then clasped with the hand and thumb can be used to move the adjuster so that the liquid starts to get inside the pipet.
4. Once the desire level is reached then the pipet is gently lifted off the liquid and placed on top of the container for liquid to be transferred.
5. In case of pipette aid, make sure that the motor is connected to a working outlet or the pipette aid is charged. Since it is automatic, there are two buttons, one for uptake of liquid in the pipet and the other for the dispensing of liquid.
Weighing and steps to making a solution
Weighing a chemical in the lab
Before you begin make sure that you are wearing appropriate personal protective equipment or PPE (usually lab coat and gloves). Sometimes you may be weighing a fine powder, then you may want to wear protective eye gear or mask, to prevent it from causing any eye, nose, or throat irritation.
Also, make sure that you know the exact amount of substance to be weighed and what weighing balance you are going to use.
Make sure to turn on the weighing balance. After you keep the weigh boat or container on the balance pan, make sure to tare the balance to zero. This will ensure that anything that you add on the weigh boat or container is reflected accurately on the digital scale. You can use spatula to add or remove the chemical.
Steps to making a solution (Weigh, mix, and store)
Method 1
- Weigh the solid chemical in a weigh boat.
- Put it in an appropriate volumetric flask.
- Add distilled water up to about 80% of the desired volume.
- Stir the solution.
- Once mixed makeup to the desired volume.
- Store it in an appropriate container.
- Autoclave if needed.
Method 2
- Weigh the solid chemical in a weigh boat.
- Put it in an appropriately sized beaker.
- Add distilled water up to about 80% of the desired volume.
- Drop a stir bar and stir the solution.
- Adjust the pH if so needed.
- Once mixed makeup to the desired volume in a graduated cylinder.
- Store it in an appropriate container.
- Autoclave if needed.
Please make sure that after you are done, you will clean the area and either keep the spatula for washing or wash it and keep it aside. Also make sure to discard the PPE to the right places.
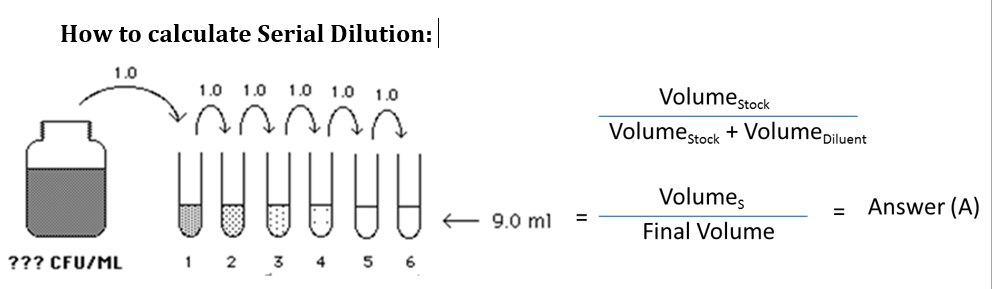
Simple and Serial Dilutions
In a simple dilution, a unit volume of a liquid from stock is combined with an appropriate volume of a solvent liquid to achieve the desired concentration.
- The dilution factor is the unit you added the +total number of unit volumes in which your material will be dissolved.
- For e.g. 1ml in 5 ml. DF= 5 folds or 1:5
A serial dilution is a series of simple dilutions which increases the dilution factor quickly.
- The source of dilution material for each step comes from the diluted
material of the previous step.
- In a serial dilution, the total dilution factor at any point is the product of the individual dilution factors in each step up to it.
Why do we need Serial Dilution?
- Example 1: A technician performed a laboratory analysis of the patient’s serum for a serum glucose (blood sugar) determination. The patient’s serum glucose was too high to read on the glucose instrument. This might require dilution.
- Example 2: 100 mg/dl solution of the substrate is needed for a laboratory procedure. All that is available is a 5000 mg/dl solution of the substrate.
- Example 3: Serial dilutions are most often used in serological procedures, where technicians need to make dilutions of a patient’s serum to determine the weakest concentration that still exhibits a reaction of some type. The RECIPROCAL of the weakest concentration exhibiting a reaction is called a “titer”.
- Example 4: If we have to measure bacteria in a wastewater sample. However as seen in the following, figure below, it is not possible to count bacteria in the first plate. It is only after a series of dilutions, that the bacteria can be counted.
Simple Dilution
Simple Dilution: A specific amount of stock is added to diluent like water to achieve a lower concentration. This is only a one-time dilution. Below you can see that each tube gets a sample from the stock.
Dilution factor means how many folds dilution.
Example 1: If its 1 ml stock in 9 ml water
Dilution Ratio = 1/ (1+9) = 1/10 or 1:10
(It is expressed as 1:Dilution Factor)
Example 2: If its 0.5 ml stock in 9.5 ml water
Dilution Ratio = 0.5/ (0.5+9.5) = 0.5/10 = 0.05 ??
(How do we convert this to 1:Dilution factor?)
There are two ways of doing this:
Also note that, if we make a dilution of 1:10 to a stock solution of 200mM, the dilution will become (1/10 x 200) 20mM.
Serial Dilution
Serial Dilution is a series of dilutions or stepwise dilutions in which the 1st dilution is from the stock, the 2nd dilution is from 1st, the 3rd dilution is from 2nd, and so on and so forth till the desired dilution factor is reached.
If V1 is added to V2 then the dilution factor at Step 1 will be
DF1= V1 / V1+V2
- The dilution factor at each step can be calculated
For e.g. at Step 3 the dilution factor (DF) = DF1 x DF2 x DF3.
- Serial dilutions find their applications in labs both clinical as well as scientific labs like determining antibody titer, diluting blood, or serum.
- Dilution factor can be popularly expressed as ratio. (Also in your experimental data)
Link: http://education.wichita.edu/saltymicro/ecology_interactives/serial_dilution.html
PRACTICE PROBLEMS: DILUTIONS
Name: Date:
- If you add 1.0 ml of a colored solution (Stock) to 4.0 ml of water in a test tube, what is the dilution ratio, of the colored solution to the final volume?
- A dilution ratio of 1:10 means there is _______ul of stock in 10 ul of final volume.
- A dilution ratio of 1:10 means ______ml of stock in 20 ml of final volume.
- A dilution ration of 1:10 means _______L of stock in 50 L of final volume.
- What is the dilution when we add 3 ml of stock to 27 ml of water?
- Find the concentrations of A, B, C, D. (Hint: Observe carefully if it is simple or serial dilution)
- A= 1ml of serum in 4ml of buffer = ____________________
- B = 1ml of A in 4 ml of buffer = _____________________
- C= 1ml of B in 4 ml of buffer = _____________________
- D= 1ml of C in 4 ml of buffer = _____________________
- If you were making a 1:10 dilution of a 100mM NaCl solution what will be its final concentration?
- If you were making 1:25 dilution of a 25mM KCl solution what will be its final concentration?
- If you have a Bradford reagent that you have to dilute 1:4. After dilution the final volume should be 20ml, how much of the Bradford reagent would you use to make this final volume (diluted solution)?
pH
pH is a unit of measure which describes the degree of acidity or alkalinity (basic) of a solution. The formal definition of pH is that it is the negative logarithm of the hydrogen ion activity.
- It is measured on a scale of 0 to 14.
- "p" means taking the negative logarithm of whatever follows in the formula. for pH, pOH, p[anything]
- pH = -log[H+]
The pH scale corresponds to the concentration of hydrogen ions.
How do we identify acids or bases?
pH 7 is considered neutral. Acid has a pH of less than 7. The base has a pH greater than 7. The lower the value, the stronger is the acid. The higher the value, the stronger is the base.
Logarithmic Scale Preview
Number Representation and Logarithms | ||
Number | Exponent/Scientific Notation | Log of the Number |
1000 | 103 | 3 |
100 | 102 | 2 |
10 | 101 | 1 |
1 | 100 | 0 |
0.1 | 10-1 | -1 |
0.01 | 10-2 | -2 |
0.001 | 10-3 | -3 |
0.0001 | 10-4 | -4 |
How do we determine pH?
Example: If an acid has an H+ concentration of 0.0001 M, find the pH.
Solution:
- First convert the number to exponential/ scientific notation, find the log, then solve the pH equation.
- H+ = 0.0001M = 10-4; log of 10-4 = -4;
- pH = - log [ H+] = - log (10-4) = - (-4) = +4 = pH
- The purpose of the negative sign in the log definition is to give a positive pH value.
Question (Solve this): If an acid has an H+ concentration of 0.000001 M, find the pH.
Example of various pH in daily life
Water undergoes auto- or self-ionization as shown in the following equation.
2 H2O --> H3O+ + OH- (What happened to H+ ions?)
- An equilibrium is established between the ions produced and the unionized water. An equilibrium expression can be written for this system.
Kw = [H3O+][OH-]
where Kw is used to represent the equilibrium constant for the ionization of water.
- The value of Kw, when experimentally measured has been determined to be 1 x 10-14.
- Mathematically, if one takes the log of both sides of the equation, remembering that when numbers are multiplied their logs are added:
log (1 x 10-14) = log [H3O+] + log [OH-]
Thus, - 14 = log[H3O+] + log [OH-]
Multiplying both sides of the equation by -1, we get
14 = - log [H3O+] - log [OH-]
14 = pH + pOH
pH Measurement
pH is measured in a number of different ways using pH paper or pH meter.
pH strips are pieces of paper that change color depending on the the acidity or alkalinity of a liquid.
pH Meter
pH Meter is an instrument to measure the hydrogen-ion activity in solutions as pH indicating its acidity or alkalinity.
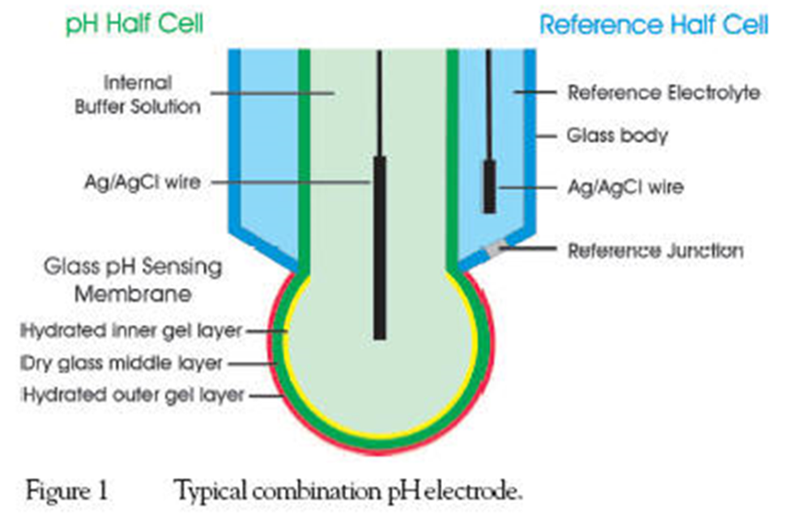
- A pH measurement system consists of three parts: a pH measuring electrode, a reference electrode, and a high input meter.
- The pH meter is a potentiometer which measures the potential developed between the glass electrode and the reference electrode.
- The pH measuring electrode is a hydrogen ion sensitive glass bulb.
- The reference electrode output does not vary with the activity of the hydrogen ion.
- In modern instruments, the two electrodes are combined into one electrode, known as a combination electrode.
How to pH
- • A sample is placed and the glass probe at the end of the retractable arm is placed in it.
- • The probe is connected to the main box.
- • There are two electrodes inside the probe that measure voltage.
- • One is contained in liquid with fixed pH. The other measures the acidity of the sample through the amount of H+ ions.
- • A voltmeter in the probe measures the difference between the voltages of the two electrodes.
- • The meter then translates the voltage difference into pH and displays it on the screen.
- • Before taking a pH measurement the meter must be calibrated using a solution of known pH.
Titration
- The process of gradually adding known amounts of reagent to a solution with which the reagent reacts while monitoring the results is called a titration.
Buffers are mixtures of weak acids and their anions (conjugate base)
- Buffers resist change in pH
- At pH = pKa, there is a 50:50 mixture of acid and anion forms of the compound
- Buffering capacity of acid/anion system is greatest at pH = pKa
- Buffering capacity is lost when the pH differs from pKa by more than 1 pH unit
pH related questions:
- If H+ ion concentration of a buffer is 10-9M, what is its pH? Is it acidic or basic?
- If the H+ concentration of a buffer is 0.0001 M, what is the pH? Is it acidic or basic?
- If the pH is 4, what is the pOH?
- If the H+ concentration is 0.00001 M, what is the OH- concentration?
- If the pH of a buffer is 7 what is the hydrogen ion concentration?
- If the pH of buffer A is 3 and pH of buffer B is 5, what is the difference in their hydrogen ion concentration?
- If the expected pH of Buffer A is 5.5 and the observed pH is 5 what is the % error in the observed as compared to the expected pH? (Percent error formula = (Theoretical - Expected)/ Theoretical x 100)
Centrifuge
CENTRIFUGATION
- A process that involves the use of centrifugal force for the separation of mixtures.
- The equipment that is used for centrifugation is called a centrifuge and the vessel that spins the sample is called rotor.
Supernatant & Pellet are separated after the process of centrifugation.
Supernatant : Liquid on top
Pellet : Solid at the bottom of the tube
Watch this video on centrifuge: https://www.youtube.com/watch?v=IhJNFGfsUus
INSTRUMENT DESIGN
Centrifugal force can be measured as:
- Revolutions per minute (RPM) or
- Acceleration applied to the sample/Relative Centrifugal force (RCF, also referred as g)
Types of Centrifuges
- Low Speed (3-6000 rpm)
- Micro centrifuge or Microfuge (Upto 12,000 rpm)
- Small amounts, less than 2 ml
- High Speed, (Upto 24,000 rpm)
- Refrigerated to counteract the build up of heat produced.
- Ultra Centrifuge (30-60,000 rpm)
- dependent upon rotor type
- Used to separate similarly sized particles
- Vacuum pressure (diffusion pump)
- Refrigerated
- Rotors are fixed angle or vertical,
- Rotors made up of heat and pressure tolerant metal
- Specialize tubes
- EX. Ti 60 = 60,000rpm limit
- Holds 8 - 38 ml tubes
Types of Rotors
- Fixed angle
- Vertical
- Horizontal/ swinging bucket
Centrifuge tubes
- Centrifuge tubes are tapered tubes of various sizes made of glass or plastic.
- They may vary in capacity from tens of mm, to much smaller capacities used in
- One of the most commonly encountered tubes is about the size and shape of a normal test tube (~ 10 cm long).
Microcentrifuge tubes
- These are used extensively in molecular biology laboratories.
- Micro-centrifuges typically accommodate micro-centrifuge tubes with capacities
- from 250 μto 2.0 ml →These are exclusively made of plastic.
Procedure of centrifugation
- Set the desired temperature
- Close the lid while temperatures are adjusting
- Check tube tops are closed
- Weigh and balance tubes
- Start when the proper temperature is achieved
- Place the tube into centrifuge in a balanced position
- Close and lock lid
- Check if speed is in RPM or RCF and accordingly set.
- Set time and brake before you begin
Balancing rotor load in a centrifuge
If there are 3 tubes, you can use a balance tube like a tube filled with water.
You should balance the tubes on the weighing scale to ensure the diagonally placed tubes have exact same weight.
If there are 3 tubes, you can use a balance tube like a tube filled with water.
You should balance the tubes on the weighing scale to ensure the diagonally placed tubes have exact same weight.
Balancing rotor load in centrifuge
Spectrophotometer
Every compound absorbs and emits lights over a certain range of wavelength. A spectrophotometer is an instrument that emits light of specific wavelength to pass through a solution and measures the transmitted light. It is used to measure the concentration of a solution, confirm chemical entity or purity.
Qualitative Analysis
- Spectrophotometer can help identify the samples that is being measured including RNA, DNA and proteins
- Purity of DNA or protein can be measured using a spectrophotometer.
Quantitative Analysis can help identify
- Proteins by colorimetric (broad wavelength analysis)
- Nucleic acids by absorption analysis (260-280nm)
- Cell Density by turbidity analysis
Three types of spectrum can be used in spectrophotometer:
Photometric Assay
- Direct measurement of the absorption of a sample at a given wavelength.
- Indirect measurement of an enzymatic reaction product that serves as an indicator of absorption that is directly proportional to absorption of the target compound. ( Ex Kinetic assays, Lowry’s assay)
Absorbance measures how much of an incident light is absorbed when it travels in a material while transmittance measures how much of the light is transmitted.
Spectrophotometer is based on the Beer-Lambert Law which states that the amount of light absorbed is directly proportional to the concentration of the solute in the solution and thickness of the solution under analysis.
Beer-Lambert law
The Beer-Lambert law states that the quantity of light absorbed by a substance dissolved in a fully transmitting solvent is directly proportional to the concentration of the substance and the path length of the light through the solution
The Beer-Lambert law also referred to as Beer’s Law, describes the relationship among absorbance, the molar solute concentration, and the length of the path the light takes to get to the sample in centimeters.
Absorbance is directly proportional to concentration and length:
A = εcl
ε is the wavelength-dependent molar absorptivity coefficient
A - Absorbance (A)
M (c) - Molar solute concentration in M (c)
l - length of the path the light takes to get to the sample in centimeters (l)
Transmittance is the relationship between the amount of light that is transmitted to the detector once it has passed through the sample (I) and the original amount of light (Io). This is expressed in the following formula. T = I / Io
Spectrophotometer Components |
Video - Inside a spectrophotometer
Spectrophotometer components
- l selector: Usually a prism, diffraction grating or set of screening filters where a specific l of monochromatic light is selectively generated.
- Slit: Regulates intensity
- Photocell: Detects absorption by using electrons in the refracted light to generate an electric current that can be amplified and measured for an absorbance value
- Absorbance scale – exponential (0-infinity) Proportion of light absorbed in solution.
- Transmission – Percentage, linear (0-100) Proportion of light passing through the solution.
- OD- Optical density
Inside our Spectrophotometer
Procedure
- Turn on the instrument. Allow it to warm up for 5 or more minutes.
- Insert the control/ blank solution into the well.
- Be sure that the sides of the cuvette are clean and dry. Set the absorbance to zero.
- Remove blank. Close the well, set the transmission to 0%.
- Now insert the sample tube and note down the reading. Toggle between Abs and %T to see absorbance and transmittance.
Absorbance & Transmittance are inversely related.
Absorbance scale – Exponential (0- Infinity)
Transmission (%T) - Percent, Linear (0-100); the proportion of light passing through a solution.
Optical Density (OD) - is measured or corrected against a blank control.
Chromatography
Chromatography refers to a family of separation techniques used to separate molecules from a mixture. Chromatography is used to separate & identify components of a mix. (Like separate individual colors. (Video: https://www.youtube.com/watch?v=08YMBGS1pYU)
In chromatography, components to be separated are distributed between two phases the stationary phase and the mobile phase.
- Mobile phase: Phase in which sample is dissolved, it may be gas, liquid.
- Stationary phase: Phase through which mobile phase is forced through like Column or TLC. How does it happen?
- Individual sample components move through the mixture in mobile phase at different speed based on their solubility.
- Molecules that spend most of their time in the mobile phase are carried along faster.
Separation of components of ink using paper chromatography
How are the mobile & stationary phases chosen in chromatography?
Mobile and stationary phases are chosen so the analyte (ink mixture to be analyzed) will distribute itself between the two phases.
Uses of Chromatography
Chromatography is a technique to
- Analyze & separate mixtures of compounds
- Identify unknown compounds
- Purify or establish the purity or concentration of compounds
- Quantify and Monitor product formation in the pharmaceutical and biotechnology industries
Chromatography is widely used by forensic teams to analyze blood and urine samples for drugs, paint analysis, and testing for the presence of explosives.
Chromatography has applications in various fields including:
- Pharmaceutical Company – determine the amount of each chemical found in new product
- Hospital – detect blood or alcohol levels in a patient’s bloodstream.
- Law Enforcement – to compare a sample found at a crime scene to samples from suspects
- Environmental Agency – determine the level of pollutants in the water supply
- Manufacturing Plant – to purify a chemical needed to make a product
Separation in Chromatography
Separation in chromatography occurs based on differences in factors such as:
- Polarity (Extent of separation of charge, positive & negative)
- Ionic strength (Separation is based on net surface charge)
- Size (Separation is based on the size of the molecule)
- Affinity (Separation is based on biological affinity such as Antigen-Antibody interaction)
There is differential interaction of molecules between an immobile (Stationary/ Solid) and a mobile (solvent) phase.
Mobile phase or Solvent phase is also known as the elution buffer in many types of chromatography.
Important terms used here: Stationary/ Immobile/ Solid phase; Mobile/ Solvent phase.
Chromatography – Types
- Partition: Partitioning the molecule of interest between an immobile (stationary) phase (column material) and a mobile phase (elution buffer). The molecule does not attach to the stationary phase.
- Adsorption: Separation by the strength of binding of a molecule of interest to the immobile phase (solid stationary phase)
Partition Chromatography
- The molecule to be separated must be dissolved in the solvent.
- Any molecule that differs in solubility in the mobile and immobile phases can be separated. Examples of Partition chromatography are:
- PC (paper chromatography)
- TLC (thin layer chromatography)
- Gel Filtration Chromatography
Adsorption Chromatography
- Separation is by the strength of binding of a molecule of interest to the immobile phase (solid stationary phase).
This is different from partition chromatography because in partition, molecules distribute themselves between mobile and stationary phases without binding but in adsorption, there is binding due to attraction between a molecule and stationary phase. The interaction of a molecule with the column material causes separation
Examples of Adsorption chromatography are:
- Ion exchange
- Affinity Chromatography
Paper Chromatography and Thin Layer Chromatography (TLC)
Two types of separation based on sample movement are possible in paper chromatography:
Ascending PC (capillary) action (More common)
- Ascending means “Going Up”
- Descending PC (gravity & capillary) interactions
- Descending means “Going Down
- Descending means “Going Down
Ascending Chromatography Descending Chromatography
Thin Layer Chromatography (TLC) is a method for identifying substances and testing the purity of compounds. TLC is a useful technique because it is relatively quick and requires small quantities of material.
The stationary phase is a thin layer of adsorbent (usually silica gel or alumina) coated on a plate.
The mobile phase is a developing liquid that travels up the stationary phase, carrying the samples with it.
Components of the samples will separate on the stationary phase according to how much they dissolve in the mobile phase and how much they adsorb on the stationary phase.
TLC Chromatogram shown in the figure below was taken after experimenting with black ink from the permanent marker (Stabilo OHPen universal) and ethanol + water mixture as solvent
Separation of black ink on TLC plate. (Photo Source: Wikipedia.)
Checkout this Youtube Video https://www.youtube.com/watch?v=CmHFVxTxkGs
Can you tell which color is most soluble and which is the least soluble?
How to set up TLC?
Which color is more soluble? (Hint: The one that is running faster or higher)
Paper or Thin Layer Chromatography – Lab Math Ratio of Fronts (Rf)
How to calculate Rf
- Mark origin at the point of start
- Let the separation of colors be completed
- Stop the run when the solvent has reached about 75-80%
- Now calculate two distances as mentioned below
Rf = Distance (Start to Center of Spot) Distance (Start to Solvent front)
Distance traveled by A is 5 cm Distance traveled by B is 2 cm
Distance traveled by the solvent front is 10cm
Rf of A = Distance traveled by A / Distance travelled by Solvent front = 5 cm/ 10 cm = 0.5
Can you calculate Rf of B?
Rf of B = Distance traveled by B / Distance traveled by Solvent front
Properties of Ratio of Fronts
Is the Rf same if compound run alone or with mixture?
Yes, because the movement of a compound depends on how soluble it is not if it is run alone or in a mixture.
Is the Rf same or different for same compound run in different solvents?
If you run the same compound in water or ethanol its solubility may be different, so its Rf is different.
Interpreting Chromatography data
- The Rf (retention factor) value for each spot should be calculated. Remember when calculating its from origin to middle of the spot not beginning or end of spot.
- It is characteristic for any given compound on the same stationary phase using the same mobile phase for development of the plates. So if you run it in anywhere in world you will always get the same Rf factor.
- Hence, known Rf values can be compared to those of unknown substances to aid in their identifications.
- In addition, the purity of a sample may be estimated from the chromatogram.
- An impure sample will often develop as two or more spots, while a pure sample will show only one spot.
What determines the separation?
- Solubility
- Dissociation constant
- Molecular size
- Temperature/humidity
- Spot size/dry spot
Solubility
- Few substances have identical solubility in any one solvent, so this can help separate components of a mixture.
- More soluble a molecule the faster it moves
Dissociation Constant
- Determines if a molecule is charged or uncharged at a certain pH,
- Why is Dissociation constant important? As this affects the solubility.
- Generally, uncharged mole separates in organic (hydrophobic) solvent.
- Charged mole separate in aqueous (hydrophilic) solvent.
Molecular Size
- Increased mole weight = more lipophilic
- Number of polar side groups (peptides) = more aqueous
Temperature and Humidity
- The saturation of solvent changes the flow rate.
- Increased temperature= increased vapor in the chamber, so it can affect the flowrate of solvent
- Increased humidity paper saturates before entering the chamber.
Spot Size
- You should always use small spot as large spot means streaking, tailing, run together with another sample
Column chromatography
The stationary phase is held in a narrow tube (not a paper) through which the mobile phase is forced under pressure or under the effect of gravity.
There are several different types of column chromatography of which 3 are mentioned here:
- Gel Filtration (Based on molecule size)
- Ion Exchange (Based on molecular charge)
- Affinity Chromatography
Gel Filtration Chromatography
Separation in Gel filtration chromatography is based on molecular size.
- Sample: The mixture you are separating in gel filtration column is made up of molecules of different sizes.
- The column is made up of beads that are like little wire cages. These beads could be:
- Dextran, Agarose, Polyacrylamide (Bio gel), Sephadex (cross-linked dextran)
The mixture being separated here is made up of two molecules: Blue (big) and Red (Small). Observe which comes out first and why?
The sample mixture being separated here is made up of two molecules: Blue (big) and Red (Small). Observe which comes out first and why?
Big molecules pass around the column beads in empty spaces and come out first.
Small molecules get stuck in the beads as the beads are like empty cages. Since the smaller molecules keep getting stuck in the column beads along the path, they take a longer time to come out in comparison to the bigger molecules from the column.
This type of chromatography is also known as:
- Size Exclusion Chromatography (SEC)
- Molecular Exclusion Chromatography (MEC)
- Molecular Sieve Chromatography (MSC)
- Gel Filtration Chromatography (GFC)
- Gel Chromatography.
Ion Exchange Chromatography
Separation based on the strength of binding to the immobile phase, based on ionic interactions.
- Anion Exchanger is positively charged column that binds to anions/ negatively charged protein.
- Cation Exchanger is negatively charged column that binds to cations/ positively charged protein.
EX: (-) Carboxy-methyl groups (CM) - Attracts positive ions (+)
Diethylaminoethyl (DEAE) - Attracts negative ions
Affinity Chromatography
Separation based on specific binding to the immobile phase by exploiting known selectivity/ affinity/interaction of certain biological molecules.
- EX: Specific binding of substrates
- Inhibitors to cofactors (NZ)
- Base sequence interactions (DNA, RNA)
- Receptors to hormones
- Antibodies to lectins
Chromatography columns we just studied are used on specific instrumentation:
- HPLC: High Pressure Liquid Chromatography
- FPLC: Fast Protein Liquid Chromatography
High-performance liquid chromatography (HPLC; formerly referred to as high-pressure liquid chromatography), is a technique in analytical chemistry used to separate, identify, and quantify each component in a mixture. It relies on pumps to pass a pressurized liquid solvent containing the sample mixture through a column filled with a solid adsorbent material.
A chromatogram is a paper printout or graph of chromatographic data. Features of a chromatogram are:
- position of peaks (x-axis) Time/ Tube #
- width of peak
- symmetry of peaks
- Height of peak (y-axis)- Absorbance
On-column detection for colored or fluorescent compounds directly after developing the chromatogram.
Monitoring of eluted fractions (PC or TLC).
Using special detectors connected to the, UV detectors, etc.