- Author:
- Sneha Narayanan
- Subject:
- Chemistry
- Material Type:
- Diagram/Illustration
- Level:
- Community College / Lower Division, College / Upper Division
- Tags:
- License:
- Creative Commons Attribution
- Language:
- English
Concept of Alkene(MOP of preparation and reaction
Overview
Concept of Alkene (Mop of preparation and reaction
A few methods of preparation of alkenes have been detailed in this article
Concept of Alkene(MOP of preparation and reaction )
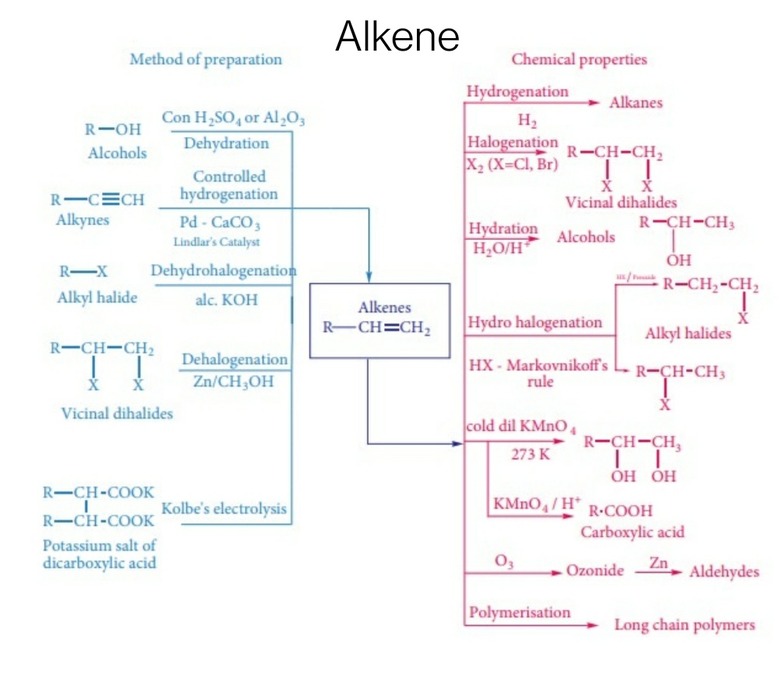
Alkenes belong to the family of hydrocarbons. They contain at least one double bond between two adjacent carbon atoms. The general chemical formula of an alkene is CnH2n. Alkenes can be prepared via various methods. A few methods of preparation of alkenes have been detailed in this article.
From alkynes: Alkynes can be used for the preparation of alkenes. Alkyne to alkene conversion is carried out by the reduction of alkynes with hydrogen in the presence of palladised charcoal. The charcoal used is moderately deactivated with the help of quinoline or sulphur compounds. This reaction results in the formation of alkenes. Palladised charcoal which is halfway deactivated is called as Lindlar’s catalyst. The alkenes obtained from the above reaction have cis geometry. In order to form trans alkenes, alkynes are made to undergo reduction with sodium in liquid ammonia.
Preparation from Alkynes
From alkyl halides: Alkenes are obtained by heating alkyl halides with alcoholic potash. Alcoholic potash is obtained by dissolving potassium hydroxide in alcohol. In this reaction, dehydrohalogenation takes place i.e. a single molecule of halogen acid is removed. The rate of reaction depends upon the alkyl group and the nature of the halogen group attached.
Preparation of Akenes from Alkyl Halides
From vicinal halides: Vicinal dihalides can be defined as the dihalides in which two adjacent carbon atoms are attached to two halogens. When such dihalides react with zinc metal, they lose halogen molecules which result in the formation of alkenes. Such a reaction of preparation of alkenes from Vicinal dihalides is known as dehalogenation.
Preparation of Alkenes from Vicinal Halides
From alcohols: Alcohols react with concentrated sulphuric acid which results in the formation of alkenes due to the elimination of a water molecule. As water molecule is removed in this reaction, it is called as acidic dehydration of alcohol and the dehydrating agent is concentrated sulphuric acid.