- Author:
- Sethabile Dladla
- Subject:
- Physical Science
- Material Type:
- Activity/Lab, Homework/Assignment, Lecture Notes
- Level:
- High School
- Tags:
- License:
- Public Domain Dedication
- Language:
- English
- Media Formats:
- Downloadable docs, Interactive, Video
Education Standards
acids worksheet
electric
Electric Circuits
Electric circuits Memorandum
electrochemical cells
Electrochemistry
memo electric
Memorandum
Projectile motion
Projectile motion exercises
Physical Sciences

Overview
The content on this pages is intented for grade 12 learners from South Africa. The curriculum standards used are those of the Curriculum Assessments Policy Standards which is a the Curriculum document used in South Africa.
Introduction to Physical Sciences (Grade 12)
INTRODUCTION TO PHYSICAL SCIENCES
WHAT IS PHYSICAL SCIENCES?
Physical Sciences investigate physical and chemical phenomena. This is done through scientific inquiry, application
of scientific models, theories and laws in order to explain and predict events in the physical environment.
This subject also deals with society’s need to understand how the physical environment works in order to benefit from
it and responsibly care for it. All scientific and technological knowledge, including Indigenous Knowledge Systems
(IKS), is used to address challenges facing society. Indigenous knowledge is knowledge that communities have held,
used or are still using; this knowledge has been passed on through generations and has been a source of many
innovations and developments including scientific developments. Some concepts found in Indigenous Knowledge
Systems lend themselves to explanation using the scientific method while other concepts do not; this is still knowledge
however.
NOTE: Reference is made using the caps document ( the South African curriculum Document)
TABLE OF CONTENTS
Acids and Bases
LESSON OBJECTIVES:
- Properties of acids and bases
- Arrhenius Theory of acids and bases
- Bronsted-Lowry acids and bases
- Conjugate acids-base pairs
- Reactions of acids and bases
- The Ph scale
- Calculating ph of strong bases
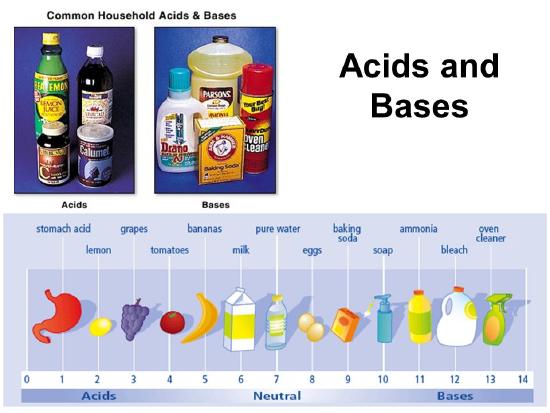
Properties of acids
- Sour to taste
- Change litmus paper from blue to red
- Conduct electricity in aqueous solutions
- Decrease ph of the solution.
Examples
Citric acid(lemons)
Vinegar
Lactic acid( milk)
Properties of bases
- Bitter to taste
- Change red litmus pare to blue
- Feels soapy or slippery
- Conducts electricity in aqueous solutions
- Increase the ph of a solution.
Examples
Limestone

Arrhenius acids and bases
Arrhenius acid
A substance which produces H+/H3O+ ons in an aqueous solution.
e.g
• HCℓ → H+ + Cℓ-
• HCℓ + H2O → H3O+ + Cℓ-
Arrhenius base
A substance which produces OH- ons in an aqueous solution.
e.g
• NaOH → Na++ + OH-−

Bronsted-Lowry acids and bases
Bronsted-lowry Acid
A substance which donates a proton.
• HCℓ + H2O → H3O+ + Cℓ−
• NH4++ H2O → H3O+ + NH3
Bronsted-lowry Base
A substance which accepts a proton.
• OH- + H+ → H2O
• NH3 + H2O → OH- + NH4

Conjugate acids
When an acid donates a proton (H+), a conjugate base is produced.
• The acid and its conjugate base are called a conjugate acid-base pair.
Examples:
HCℓ ⇌ H+ + Cℓ-
Acid conjugate base
H2SO4 ⇌ H+ + HSO4-
acid conjugate base
Conjugate base
Vertical projectile motion
LESSON OBJECTIVES
•Introduction to vertical projectile motion
•Factors affecting vertical projectile motion.
•Velocity
•Acceleration vs displacement.
•Explanation when the object is dropped from rest
•Explanation when the object is thrown vertically upward
What is a projectile motion?
In simpler way when an object is projected either upward or downward, the only force acting on is gravity.

Factors affecting free falling objects
Objects that are projected either upward or downward do not encounter air resistance. This means that the air resistance is negligible.
All objects that are under a free fall on Earth accelerate downwards at a rate of 9.8m/s2.
Velocity
- The magnitude and direction of velocity changes.
- When it is moving upward
- The magnitude decreases and its direction is upward.
- When it is moving down
- The magnitude increases and the direction is downward.
- At the top just as the object turns to go down the velocity becomes zero.
Figure 2: showing the different magnitudes of velocity as it is projected.
Equations of motion
Electric circuits
LESSON OBJECTIVES:
Ohms law
Revision of series circuits
Revision of parallel circuits
Internal resistance
Past examination papers
Ohms law
Ohm's law states that the current through a conductor between two points is directly proportional to the voltage across the two points.
Series circuits
The defining characteristic of a series circuit is that there is only one path for current to flow.

Characteristics of series circuits
- Current: The amount of current is the same through any component in a series circuit.
- Resistance: The total resistance of any series circuit is equal to the sum of the individual resistances.
- Voltage: The supply voltage in a series circuit is equal to the sum of the individual voltage drop.

Parallel circuits
The basic idea of a “parallel” connection, on the other hand, is that all components are connected across each other’s leads.

Characteristics of the parallel circuits
- Voltage: Voltage is equal across all components in a parallel circuit.
- Current: The total circuit current is equal to the sum of the individual branch currents.
- Resistance: Individual resistances diminish to equal a smaller total resistance rather than add to make the total.

Examples
Electrochemical cells
LESSON OBJECTIVES
Introduction to electrochemistry
Vocabulary
Redox reaction: reduction and oxidation
Electrochemical cell: electrolytic cell and galvanic/voltaic cell.
What is electrochemistry?
It refers to chemical reactions during which chemical energy is converted to electric energy, or electric energy is converted to chemical energy.
There are two types of electrochemical cells:
Electrolytic cell: a cell in which electrical energy is converted into chemical energy.
Galvanic (voltaic) cell: a cell in which chemical energy is converted into electrical energy.

Characteristics of an Electrolytic cell
Uses electricity to create chemical reactions
Does not have a salt bridge.
Non-spontaneous redox reactions.
Endothermic reactions.
Emf is always negative.

Characteristics of a Galvanic cell
It uses chemical reactions to create electricity.
Spontaneous reaction.
Has a salt bridge.
Exothermic reaction.
Emf is always positive.

