- Author:
- Anna McCollum, Amanda Spangler, Jillian Gorrell, ALka Sharma, Madonna Kemp
- Subject:
- Life Science
- Material Type:
- Full Course
- Level:
- College / Upper Division
- Tags:
- License:
- Creative Commons Attribution
- Language:
- English
- Media Formats:
- Graphics/Photos
1.3 Breakdown of Pryuvate and Krebs Cycle
1.4 Electron Transport Chain
1.5 Chemiosmosis and ATP Production
1.6 Anaerobic Cellular Respiration
1.7 Regulatory Mechanisms
1_Cellular-Respiration
Cellular Respiration
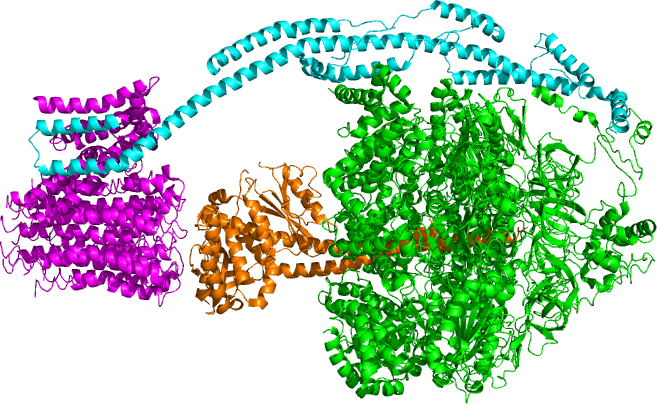
Overview
Bovine mitochondrial ATP synthase based on PDB 5ARA. The FO, F1, axle, and stator regions are color coded magenta, green, orange, and cyan respectively. Picture Created in PyMol. BiochemEkaterina, CC BY-SA 4.0 <https://creativecommons.org/licenses/by-sa/4.0>, via Wikimedia Commons
Principles of Biology by Robert Bear, David Rintoul, Bruce Snyder, Marth Smith -Caldas, Christopher Herren, Eva Horne. 2016. The textbook was originally published and is also available to download at http://cnx.org/contents/db89c8f8-a27c-4685-ad2a-19d11a2a7e2e@24.1.It is licensed under a Creative Commons Attribution License 4.0 license.
Did you have an idea for improving this content? We’d love your input.
Introduction
Learning Objectives
- Explain the process of glycolysis.
- Distinguish between aerobic respiration and fermentation.
- Explain how the Krebs cycle oxidizes pyruvate.
- Explain the fate of the electrons in the Electron Transport Chain (ETC).
- Explain why the production of ATP is called oxidative phosphorylation.
Key Terms
aerobic respiration - a process in which organisms convert energy in the presence of oxygen
ATP - adenosine triphosphate, the cell's energy currency
Beta-oxidation - a process by which fatty acids are catabolized
Cellular respiration - a process in which an organism converts energy in the presence of oxygen
Decarboxylation - removal of the carboxyl group as carbon atom or CO2
Electron transport chain - a group of proteins on the inner membrane of mitochondria that pass electrons and use the energy released by the electrons to move hydrogen ions against the concentration gradient into the intermembrane space
Fermentation - a process of regenerating NAD+ with either an inorganic or organic compound serving as the final electron acceptor; occurs in the absence of oxygen
Gluconeogenesis - the process of glucose synthesis
Glucose - a six-carbon monosaccharide
Glycolysis - the process of breaking glucose into two three-carbon molecules with the production of ATP and NADH
Krebs cycle - (also citric acid cycle; tricarboxylic acid cycle; TCA cycle) series of enzyme-catalyzed chemical reactions of central importance in all living cells for extraction of energy from carbohydrates
Metabolism - all the chemical reactions that take place inside cells, including anabolism and catabolism
Mitochondria - (singular = mitochondrion) cell organelles responsible for carrying out cellular respiration, resulting in producing ATP, the cell's main energy-carrying molecule
NADH - high-energy molecule
Oxidation - loss of electrons
Oxidative phosphorylation - production of ATP using the process of chemiosmosis in the presence of oxygen
Oxidize - cause electron loss in a substance
Pyruvate/Pyruvic acid - three-carbon sugar that can be decarboxylated and oxidized to make acetyl CoA, which enters the citric acid cycle under aerobic conditions, the end product of glycolysis

The electrical energy plant in figure 2.1.1 converts energy from one form to another form that can be more easily used. This type of generating plant starts with underground thermal energy (heat) and transforms it into electrical energy that will be transported to homes and factories. Like a generating plant, plants and animals also must take in energy from the environment and convert it into a form that their cells can use. In the process of photosynthesis, plants and other photosynthetic producers take in energy in the form of light (solar energy) and convert it into chemical energy in the form of glucose, which stores this energy in its chemical bonds. Then, a series of metabolic pathways, collectively called cellular respiration, extracts the energy from the bonds in glucose and converts it into a form that all living things can use.
Energy production within a cell involves many coordinated chemical pathways. Most of these pathways are combinations of oxidation and reduction reactions, which occur at the same time. An oxidation reaction strips an electron from an atom in a compound; the addition of this stripped electron to another compound is a reduction reaction. Because oxidation and reduction usually occur together, these pairs of reactions are called oxidation-reduction reactions, or redox reactions, figure 2.1.2.
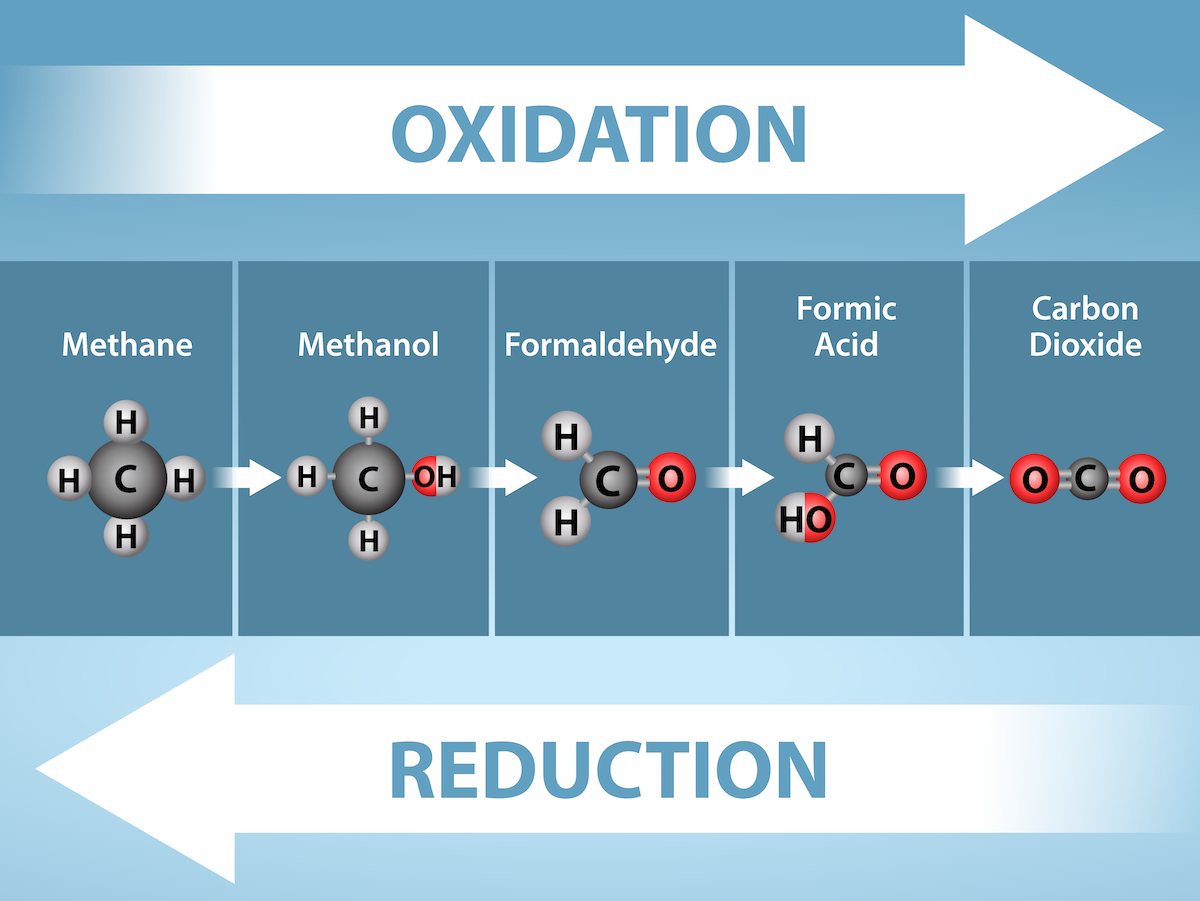
Electrons and Energy
The removal of an electron from a molecule (oxidizing it), causes a decrease in the potential energy of the oxidized compound. However, the electron (sometimes as part of a hydrogen atom) does not remain unbonded in the cytoplasm of a cell. Rather, the electron is shifted to a second compound, reducing the second compound. The shift of an electron from one compound to another removes some potential energy from the first compound (the oxidized compound) and increases the potential energy of the second compound (the reduced compound). The transfer of electrons between molecules is important because most of the energy stored in atoms and used to fuel cell functions is in the form of high-energy electrons. The transfer of energy in the form of high-energy electrons allows the cell to transfer and use energy in an incremental fashion—in small packages rather than in a single, destructive burst.
Electron Carriers
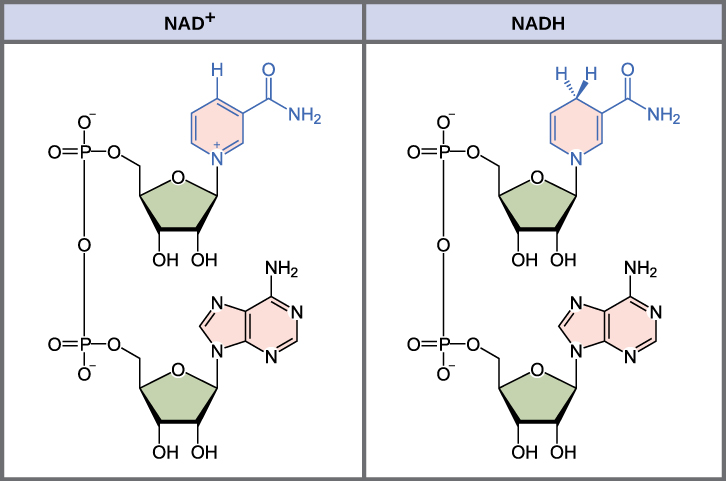
In living systems, a small class of compounds functions as electron shuttles: they bind and carry high-energy electrons between compounds in biochemical pathways. The principal electron carriers we will consider are derived from the B vitamin group and are derivatives of nucleotides. These compounds can be easily reduced (that is, they accept electrons) or oxidized (they lose electrons). Nicotinamide adenine dinucleotide (NAD) (figure 2.1.3) is derived from vitamin B3—niacin. NAD+ is the oxidized form of the molecule; NADH is the reduced form of the molecule after it has accepted two electrons and a proton, which together are the equivalent of a hydrogen atom with an extra electron. Note that if a compound has an “H” on it, it is generally reduced; for instance, NADH is the reduced form of NAD. The second variation of NAD, NADP, contains an extra phosphate group
NAD+ can accept electrons from an organic molecule according to the general equation:
RH + NAD+ ------> NADH + R
Reducing agent + Oxidizing agent -----> Reduced + Oxidized
When electrons are added to a compound, it is reduced. A compound that reduces another is called a reducing agent. In the above equation, RH is a reducing agent, and NAD+ is reduced to NADH. When electrons are removed from a compound, it is oxidized. A compound that oxidizes another is called an oxidizing agent. In the above equation, NAD+ is an oxidizing agent that grabs the H and causes RH to be oxidized to R.
Similarly, there are other electron carriers such as FAD+. Flavin adenine dinucleotide (FAD+) is derived from vitamin B2, also called riboflavin. Its reduced form is FADH2. Both NAD+ and FAD+ are extensively used in energy extraction from sugars, and NADP plays an important role in anabolic reactions and photosynthesis in plants.
ATP in Living Systems
A living cell cannot store significant amounts of free energy. Excess free energy would result in an increase of heat in the cell, which would result in excessive thermal motion that could damage and then destroy the cell. Rather, a cell must be able to handle that energy in a way that enables the cell to store energy safely and release it for use only as needed. Living cells accomplish this by using the compound adenosine triphosphate (ATP). ATP is often called the “energy currency” of the cell, and, like currency, this versatile compound can be used to fill any energy need of the cell. It can do this because it functions similarly to a rechargeable battery.
When ATP is broken down, usually by the removal of its terminal phosphate group, energy is released. The energy is used to do work by the cell, usually when the released phosphate binds to another molecule, which activates it. For example, in the mechanical work of muscle contraction, ATP supplies the energy to move the contractile muscle proteins. Recall the active transport work of the sodium-potassium pump in cell membranes. ATP alters the structure of the integral protein that functions as the pump, changing its affinity for sodium and potassium. In this way, the cell performs work, pumping ions against their electrochemical gradients.
ATP Structure and Function
At the heart of ATP is a molecule of adenosine monophosphate (AMP), which is composed of an adenine molecule bonded to a ribose molecule and to a single phosphate group (Figure 2.1.4). Ribose is a five-carbon sugar found in RNA, and AMP is one of the nucleotides in RNA. The addition of a second phosphate group to this core molecule results in the formation of adenosine diphosphate (ADP); the addition of a third phosphate group forms adenosine triphosphate (ATP).
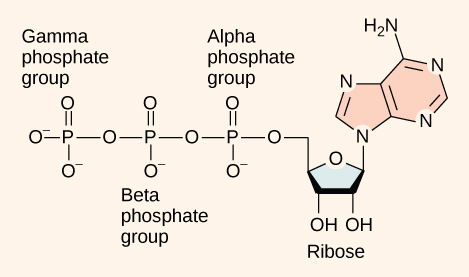
The addition of a phosphate group to a molecule requires energy. Phosphate groups are negatively charged and thus repel one another when they are arranged in series, as they are in ADP and ATP. This repulsion makes the ADP and ATP molecules inherently unstable. The release of one or two phosphate groups from ATP, a process called dephosphorylation, releases energy.
Energy from ATP
Hydrolysis is the process of breaking complex macromolecules apart. During hydrolysis, water is split, or lysed, and the resulting hydrogen atom (H+) and a hydroxyl group (OH-), or hydroxide, are added to the larger molecule. The hydrolysis of ATP produces ADP, together with an inorganic phosphate ion (Pi), and the release of free energy. To carry out life processes, ATP is continuously broken down into ADP, and like a rechargeable battery, ADP is continuously regenerated into ATP by the reattachment of a third phosphate group. Water, which was broken down into its hydrogen atom and a hydroxyl group (hydroxide) during ATP hydrolysis, is regenerated when a third phosphate is added to the ADP molecule, reforming ATP.
Obviously, energy must be infused into the system to regenerate ATP. In nearly every living thing on Earth, the energy comes from the metabolism of glucose, fructose, or galactose, all isomers with the chemical formula C6H12O6 but with different molecular configurations. In this way, ATP is a direct link between the limited set of exergonic pathways of glucose catabolism and the multitude of endergonic pathways that power living cells.
Phosphorylation
Recall that, in some chemical reactions, enzymes may bind to several substrates that react with each other on the enzyme, forming an intermediate complex. An intermediate complex is a temporary structure, and it allows one of the substrates (such as ATP) and reactants to more readily react with each other. In reactions involving ATP, ATP is one of the substrates and ADP is a product.
During an endergonic chemical reaction, ATP forms an intermediate complex with the substrate and enzyme in the reaction. This intermediate complex allows the ATP to transfer its third phosphate group, with its energy, to the substrate; this is a process called phosphorylation. Phosphorylation refers to the addition of a phosphate (~P). This is illustrated by the following generic reaction, in which A and B represent two different substrates:
A + enzyme + ATP→ [A − enzyme − ∼P] → B + enzyme + ADP + phosphate ion
When the intermediate complex breaks apart, the energy is used to modify the substrate and convert it into a product of the reaction. The ADP molecule and a free phosphate ion are released into the medium and are available for recycling through cell metabolism.
Substrate Phosphorylation
ATP is generated through two mechanisms during the breakdown of glucose. A few ATP molecules are generated (that is, regenerated from ADP) as a direct result of the chemical reactions that occur in the catabolic pathways. A phosphate group is removed from an intermediate reactant in the pathway, and the free energy of the reaction is used to add the third phosphate to an available ADP molecule, producing ATP (Figure 2.1.5). This very direct method of phosphorylation is called substrate-level phosphorylation.
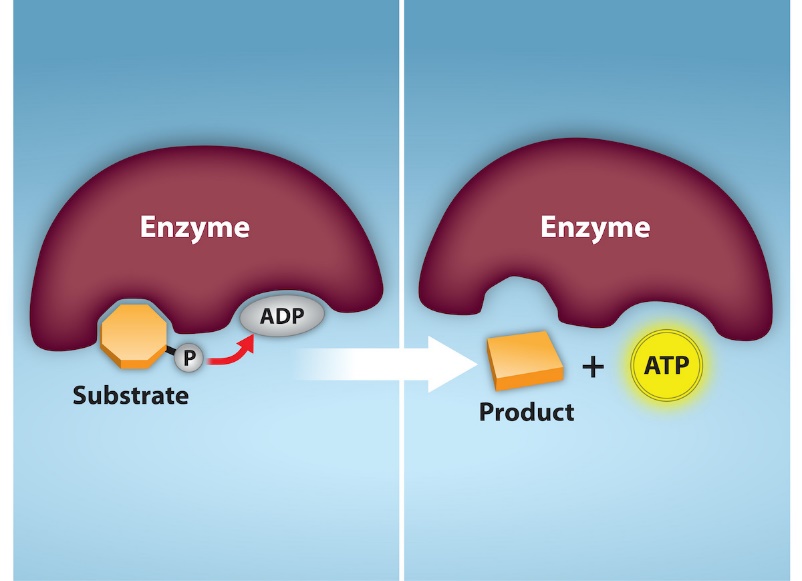
Oxidative Phosphorylation
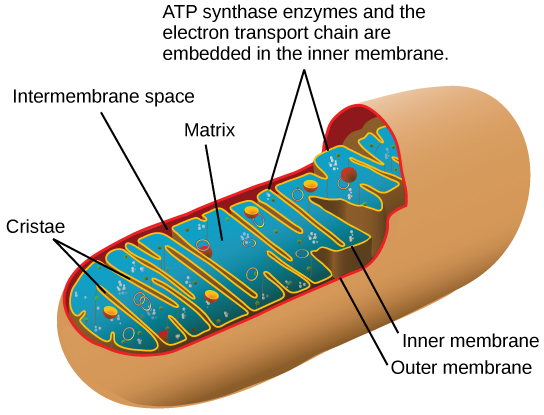
Most of the ATP generated during glucose catabolism, however, is derived from a much more complex process: chemiosmosis. Chemiosmosis, a process of ATP production in cellular metabolism, is used to generate 90 percent of the ATP made during glucose catabolism and is also the method used in the light reactions of photosynthesis to harness the energy of sunlight. Chemiosmosis takes place in mitochondria, (Figure 2.1.6) within a eukaryotic cell or the plasma membrane of a prokaryotic cell. The production of ATP using the process of chemiosmosis is called oxidative phosphorylation because of the involvement of oxygen in the process.
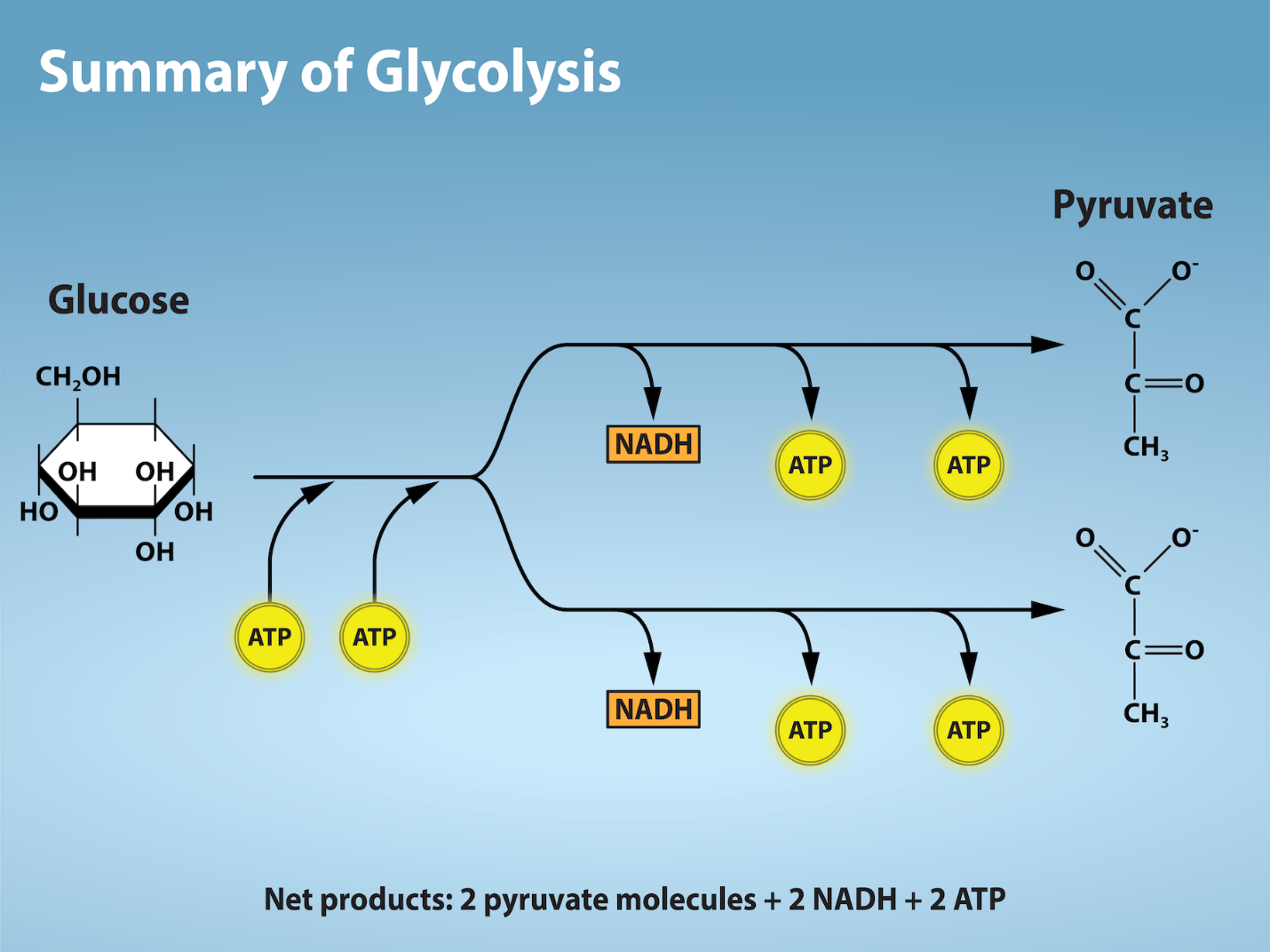
Glycolysis
As you have read, nearly all of the energy used by living cells comes to them in the bonds of a sugar molecule called glucose. Glycolysis is the first step in the breakdown of glucose to extract energy for cellular metabolism. In fact, nearly all living organisms carry out glycolysis as part of their metabolism. The process does not use oxygen directly; therefore, it is termed anaerobic. Glycolysis takes place in the cytoplasm of both prokaryotic and eukaryotic cells.
Glycolysis begins with the six-carbon ring-shaped structure of a single glucose molecule and ends with two molecules of a three-carbon sugar called pyruvate. Glycolysis consists of two distinct phases. The first part of the glycolysis pathway traps the glucose molecule in the cell and uses energy to modify it so that the six-carbon sugar molecule can be split evenly into the two three-carbon molecules. The second part of glycolysis extracts energy from the molecules and stores it in the form of ATP and NADH—the reduced form of NAD (figure 2.1.7).
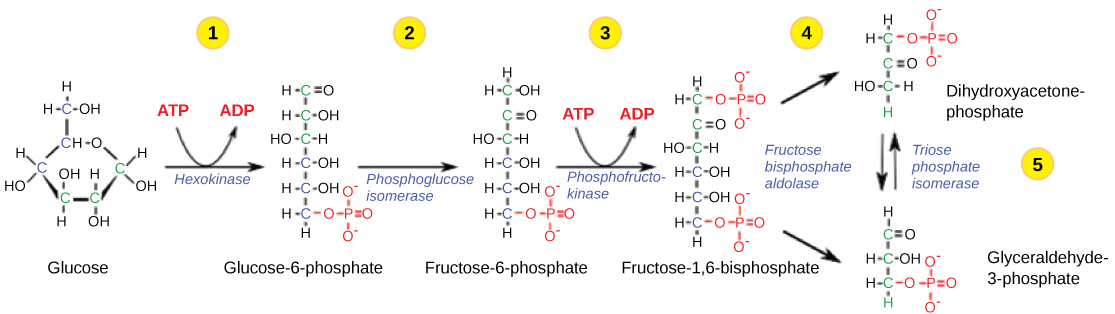
First Half of Glycolysis (Energy-Requiring Steps)
Step 1. The first step in glycolysis (Figure 2.1.8) is catalyzed by hexokinase, an enzyme with broad specificity that catalyzes the phosphorylation of six-carbon sugars. ATP is used as the source of phosphate by hexokinase to phosphorylate glucose, producing glucose-6-phosphate, a more reactive form of glucose. This reaction prevents the phosphorylated glucose molecule from leaving the cell because the negatively charged phosphate will not allow it to cross the hydrophobic interior of the plasma membrane.
Step 2. In the second step of glycolysis, an isomerase converts glucose-6-phosphate into one of its isomers: fructose-6-phosphate (this isomer has a phosphate attached at the location of the sixth carbon of the ring). An isomerase is an enzyme that catalyzes the conversion of a molecule into one of its isomers. (This conversion from phosphoglucose to phospho-fructose allows the eventual split of the sugar into two three-carbon molecules.)
Step 3. The third step is the phosphorylation of fructose-6-phosphate, catalyzed by the enzyme phosphofructokinase. A second ATP molecule donates a high-energy phosphate to fructose-6-phosphate, producing fructose-1,6-bisphosphate. In this pathway, phosphofructokinase is a rate-limiting enzyme. It is active when the concentration of ADP is high; it is less active when ADP levels are low, and the concentration of ATP is high. Thus, if there is “sufficient” ATP in the system, the pathway slows down. This is a type of end product inhibition since ATP is the end product of glucose catabolism.
Step 4. The newly added high-energy phosphates further destabilize fructose-1,6-bisphosphate. The fourth step in glycolysis employs the enzyme, aldolase, to cleave fructose-1,6-bisphosphate into two three-carbon isomers: dihydroxyacetone phosphate and glyceraldehyde-3-phosphate.
Step 5. In the fifth step, an isomerase transforms the dihydroxyacetone-phosphate into its isomer: glyceraldehyde-3-phosphate. Thus, the pathway will continue with two molecules of a glyceraldehyde-3-phosphate. At this point in the pathway, there is a net investment of energy from two ATP molecules in the breakdown of one glucose molecule.
Second Half of Glycolysis (Energy-Releasing Steps)
So far, glycolysis has cost the cell two ATP molecules and produced two small, three-carbon sugar molecules. Both of these molecules will proceed through the second half of the pathway, and sufficient energy will be extracted to pay back the two ATP molecules that were used as an initial investment, as well as produce a profit for the cell of two additional ATP molecules and two even higher-energy NADH molecules.
Step 6. The sixth step in glycolysis (Figure 2.1.9) oxidizes the sugar (glyceraldehyde-3-phosphate) by extracting high-energy electrons, which are picked up by the electron carrier NAD+, producing NADH. The sugar is then phosphorylated by the addition of a second phosphate group, producing 1,3-bisphosphoglycerate. Note that the second phosphate group does not require another ATP molecule.
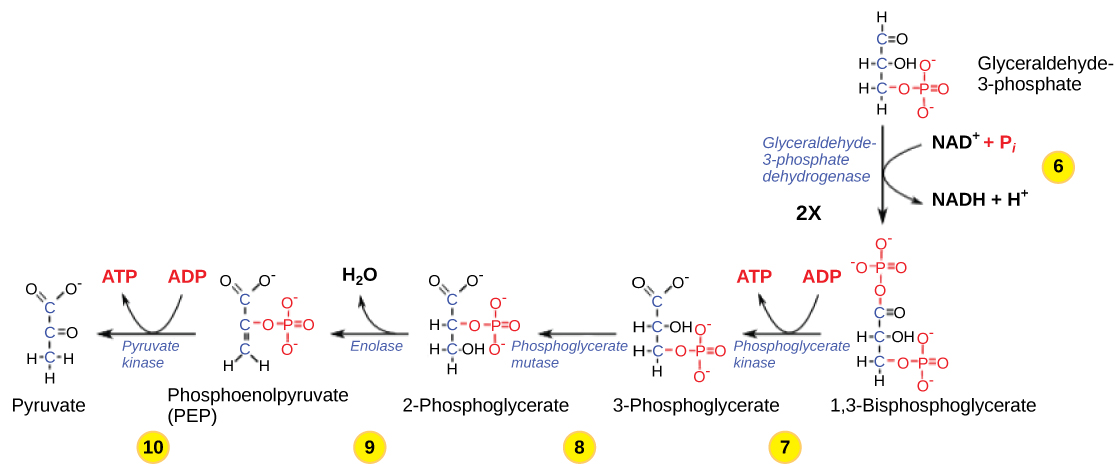
Here again, this is a potential limiting factor for this pathway. The continuation of the reaction depends upon the availability of the oxidized form of the electron carrier, NAD+. Thus, NADH must be continuously oxidized back into NAD+ in order to keep this step going. If NAD+ is not available, the second half of glycolysis slows down or stops. If oxygen is available in the system, the NADH will be oxidized readily, though indirectly, and the high-energy electrons from the hydrogen released in this process will be used to produce ATP. In an environment without oxygen, an alternate pathway (fermentation) can provide the oxidation of NADH to NAD+.
Step 7. In the seventh step, catalyzed by phosphoglycerate kinase—an enzyme named for the reverse reaction, 1,3-bisphosphoglycerate donates a high-energy phosphate to ADP, forming one molecule of ATP. (This is an example of substrate-level phosphorylation.) A carbonyl group on the 1,3-bisphosphoglycerate is then oxidized to a carboxyl group, forming 3-phosphoglycerate.
Step 8. In the eighth step, the remaining phosphate group in 3-phosphoglycerate moves from the third carbon to the second carbon, producing 2-phosphoglycerate—an isomer of 3-phosphoglycerate. The enzyme catalyzing this step is a mutase (an isomerase).
Step 9. Enolase catalyzes the ninth step. This enzyme causes 2-phosphoglycerate to lose water from its structure; this is a dehydration reaction, resulting in the formation of a double bond that increases the potential energy in the remaining phosphate bond and produces phosphoenolpyruvate (PEP).
Step 10. The last step in glycolysis is catalyzed by the enzyme pyruvate kinase (the enzyme in this case is named for the reverse reaction of pyruvate’s conversion into PEP). This step results in the production of a second ATP molecule by substrate-level phosphorylation and the compound pyruvic acid (or its salt form, pyruvate). Many enzymes in enzymatic pathways are named for the reverse reactions since the enzyme can catalyze both forward and reverse reactions; these may have been described initially by the reverse reaction that takes place in vitro, under non-physiological conditions.
Gain a better understanding of the breakdown of glucose by glycolysis by visiting this site. below.
Outcomes of Glycolysis
Glycolysis begins with glucose and produces two pyruvate molecules, four new ATP molecules, and two molecules of NADH. (Note: two ATP molecules are used in the first half of the pathway to prepare the six-carbon ring for cleavage, so the cell has a net gain of two ATP molecules and two NADH molecules for its use). If the cell cannot catabolize the pyruvate molecules further, it will harvest only two ATP molecules from one molecule of glucose. Mature mammalian red blood cells do not have mitochondria and thus are not capable of aerobic respiration—the process in which organisms convert energy in the presence of oxygen; glycolysis is their sole source of ATP. If glycolysis is interrupted, these cells lose their ability to maintain their sodium-potassium pumps and eventually die.
The last step in glycolysis will not occur if pyruvate kinase—the enzyme that catalyzes the formation of pyruvate—is not available in sufficient quantities. In this situation, the entire glycolysis pathway will proceed, but only two ATP molecules will be made in the second half. Thus, pyruvate kinase is a rate-limiting enzyme for glycolysis.
Access for free at https://openstax.org/books/biology-2e/pages/7-2-glycolysis
Breakdown of Pyruvate and Krebs Cycle
If oxygen is available, aerobic respiration will go forward. In eukaryotic cells, the pyruvate molecules produced at the end of glycolysis are transported into the mitochondria, which are the sites of cellular respiration. There, pyruvate is transformed into an acetyl group that will be picked up and activated by a carrier compound called coenzyme A (CoA). The resulting compound is called acetyl CoA. CoA is derived from vitamin B5, pantothenic acid. Acetyl CoA can be used in a variety of ways by the cell, but its major function is to deliver the acetyl group derived from pyruvate to the next stage of the pathway in glucose catabolism. In order for pyruvate the product of glycolysis—to enter the next pathway, it must undergo several changes. The conversion is a three-step process (Figure 2.1.10).
Step 1. A carboxyl group is removed from pyruvate, releasing a molecule of carbon dioxide into the surrounding medium. This reaction creates a two-carbon hydroxyethyl group bound to the enzyme (pyruvate dehydrogenase). We should note that this is the first of the six carbons from the original glucose molecule to be removed. (This step proceeds twice because there are two pyruvate molecules produced at the end of glycolysis for every molecule of glucose metabolized anaerobically; thus, two of the six carbons will have been removed at the end of both steps.)
Step 2. The hydroxyethyl group is oxidized to an acetyl group, and the electrons are picked up by NAD+, forming NADH. The high-energy electrons from NADH will be used later to generate ATP.
Step 3. The enzyme-bound acetyl group is transferred to CoA, producing a molecule of acetyl CoA.
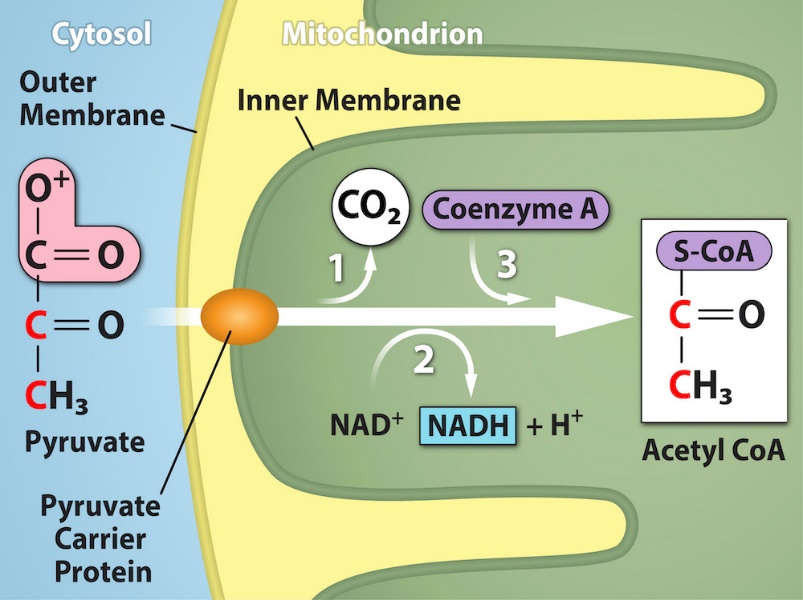
Note that during the second stage of glucose metabolism, whenever a carbon atom is removed, it is bound to two oxygen atoms, producing carbon dioxide—one of the major end products of cellular respiration.
Acetyl CoA to CO2
In the presence of oxygen, acetyl CoA delivers its acetyl (2C) group to a four-carbon molecule known as oxaloacetate in order to form citrate—a six-carbon molecule with three carboxyl groups; this pathway will harvest the remainder of the extractable energy from what began as a glucose molecule and release the remaining four CO2 molecules. This single pathway is called different names: the citric acid cycle (named after the first intermediate formed), the TCA cycle (because citric acid or citrate and isocitrate are tricarboxylic acids), and the Krebs cycle (after Hans Krebs, who first identified the steps in the pathway in the 1930s in pigeon flight muscles).
Citric Acid Cycle (Krebs Cycle)
Like the conversion of pyruvate to acetyl CoA, the citric acid cycle takes place in the matrix of mitochondria. Almost all of the enzymes of the citric acid cycle are soluble, with the single exception of the enzyme succinate dehydrogenase, which is embedded in the inner membrane of the mitochondrion. Unlike glycolysis, the citric acid cycle is a closed-loop: the last part of the pathway regenerates the compound used in the first step. The eight steps of the cycle are a series of redox, dehydration, hydration, and decarboxylation reactions that produce two carbon dioxide molecules, one GTP/ATP, and the reduced carriers NADH and FADH2 (Figure 2.1.11). This is considered an aerobic pathway because the NADH and FADH2 produced must transfer their electrons to the next pathway in the system, which will use oxygen. If this transfer does not occur, the oxidation steps of the citric acid cycle also do not occur. Note that the citric acid cycle produces very little ATP directly and does not directly consume oxygen.
Steps in the Citric Acid Cycle
Step 1. Prior to the first step, a transitional phase occurs during which pyruvic acid is converted to acetyl CoA. Then, the first step of the cycle begins: This condensation step combines the two-carbon acetyl group with a four-carbon oxaloacetate molecule to form a six-carbon molecule of citrate. CoA is bound to a sulfhydryl group (-SH) and diffuses away to eventually combine with another acetyl group. This step is irreversible because it is highly exergonic. The rate of this reaction is controlled by negative feedback and the amount of ATP available. If ATP levels increase, the rate of this reaction decreases. If ATP is in short supply, the rate increases.
Step 2. In step two, citrate loses one water molecule and gains another as citrate is converted into its isomer, isocitrate.
Step 3. In step three, isocitrate is oxidized, producing a five-carbon molecule, α-ketoglutarate, along with a molecule of CO2 and two electrons, which reduce NAD+ to NADH. This step is also regulated by negative feedback from ATP and NADH and a positive effect of ADP.
Step 4. Steps three and four are both oxidation and decarboxylation steps, which as we have seen, release electrons that reduce NAD+ to NADH and release carboxyl groups that form CO2 molecules. Alpha-ketoglutarate is the product of step three, and a succinyl group is the product of step four. CoA binds with the succinyl group to form succinyl CoA. The enzyme that catalyzes step four is regulated by feedback inhibition of ATP, succinyl CoA, and NADH.
Step 5. In step five, a carboxyl group is substituted for coenzyme A, producing a high-energy bond. This energy is used in substrate-level phosphorylation (during the conversion of the succinyl group to succinate) to form either guanine triphosphate (GTP) or ATP. There are two possible forms of the enzyme—called isoenzymes—that may result during this step, depending upon the type of animal tissue in which they are found. One form is found in tissues that use large amounts of ATP, such as the heart and skeletal muscle; this form produces ATP. The second form of the enzyme is found in tissues that have a high number of anabolic pathways, such as the liver; this form produces GTP. GTP is energetically equivalent to ATP; however, its use is more restricted. In particular, protein synthesis primarily uses GTP.
Step 6. Step six is a dehydration process that converts succinate into fumarate. Two hydrogen atoms are transferred to FAD, reducing it to FADH2. (Note: the energy contained in the electrons of these hydrogens is insufficient to reduce NAD+ but adequate to reduce FAD.) Unlike NADH, this carrier remains attached to the enzyme and transfers the electrons to the electron transport chain directly. This process is made possible by the localization of the enzyme catalyzing this step inside the inner membrane of the mitochondrion.
Step 7. Water is added by hydrolysis to fumarate during step seven, and malate is produced. The last step in the citric acid cycle regenerates oxaloacetate by oxidizing malate. Another molecule of NADH is then produced in the process.
Link to learning
View an animation of the citric acid cycle here.
Products of the Citric Acid Cycle
Two carbon atoms come into the citric acid cycle from each acetyl group, representing four out of the six carbons of one glucose molecule. Two carbon dioxide molecules are released at each turn of the cycle; however, these do not necessarily contain the most recently added carbon atoms. The two acetyl carbon atoms will eventually be released during later turns of the cycle; thus, all six carbon atoms from the original glucose molecule are eventually incorporated into carbon dioxide. Each turn of the cycle forms three NADH molecules and one FADH2 molecule. These carriers will connect with the last portion of aerobic respiration, the electron transport chain, to produce ATP molecules. One GTP or ATP is also made in each cycle. Several of the intermediate compounds in the citric acid cycle can be used in synthesizing nonessential amino acids; therefore, the cycle is amphibolic (both catabolic and anabolic).
Access for free at https://openstax.org/books/biology-2e/pages/7-3-oxidation-of-pyruvate-and-the-citric-acid-cycle
Electron Transport Chain
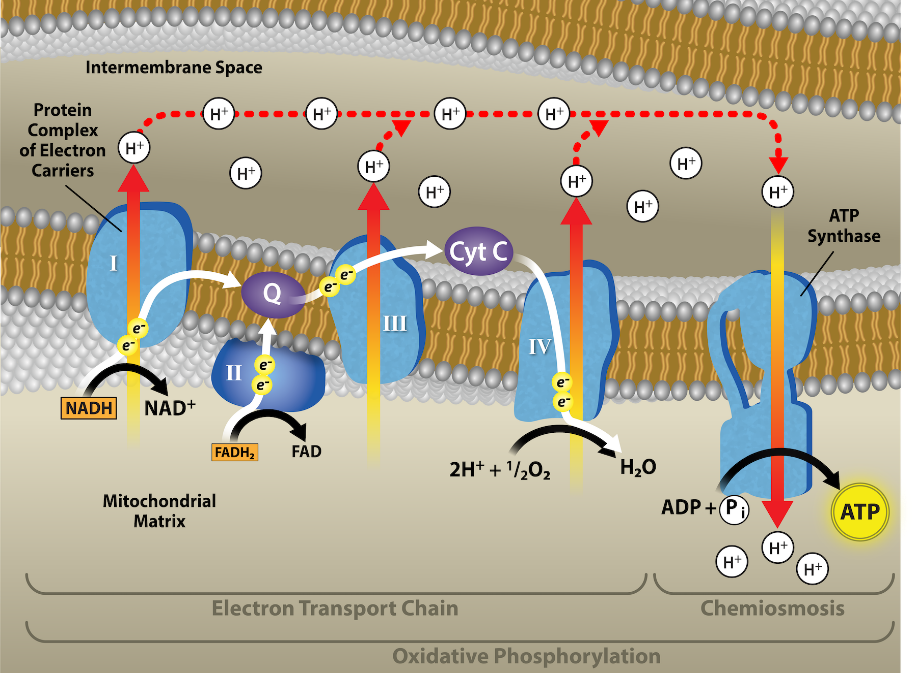
You have just read about two pathways in glucose catabolism—glycolysis and the citric acid cycle—that generate ATP. Most of the ATP generated during the aerobic catabolism of glucose, however, is not generated directly from these pathways. Instead, it is derived from a process that begins by moving electrons through a series of electron carriers that undergo redox reactions. This process causes hydrogen ions to accumulate within the intermembranous space. Therefore, a concentration gradient forms in which hydrogen ions diffuse out of the intermembranous space into the mitochondrial matrix by passing through ATP synthase. The current of hydrogen ions powers the catalytic action of ATP synthase, which phosphorylates ADP, producing ATP.
The electron transport chain (Figure 2.1.12) is the last component of aerobic respiration and is the only part of glucose metabolism that uses atmospheric oxygen. Oxygen continuously diffuses into plant tissues (typically through stomata), as well as into fungi and bacteria; however, in animals, oxygen enters the body through a variety of respiratory systems. Electron transport is a series of redox reactions that resembles a relay race or bucket brigade in that electrons are passed rapidly from one component to the next, to the endpoint of the chain where the electrons reduce molecular oxygen and, along with associated protons, produce water. There are four complexes composed of proteins, labeled I through IV in Figure 2.1.12. The aggregation of these four complexes, together with associated mobile, accessory electron carriers, is called the electron transport chain. The electron transport chain is present with multiple copies in the inner mitochondrial membrane of eukaryotes and within the plasma membrane of prokaryotes.
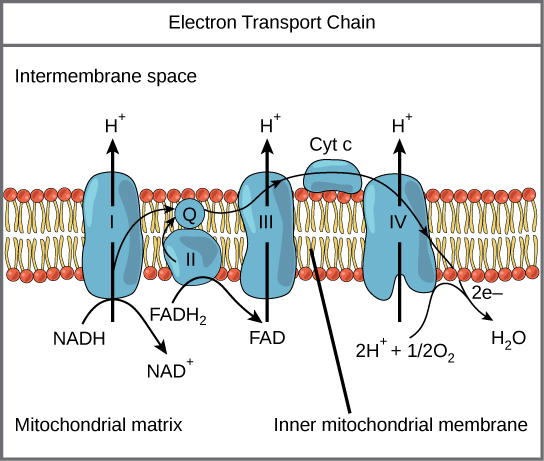
Complex I
First, two electrons are carried to the first complex via NADH. This complex, labeled I, is composed of flavin mononucleotide (FMN) and an iron-sulfur (Fe-S)-containing protein. FMN, which is derived from vitamin B2—riboflavin—is one of several prosthetic groups or cofactors in the electron transport chain. A prosthetic group is a nonprotein molecule required for the activity of a protein. Prosthetic groups are organic or inorganic, nonpeptide molecules bound to a protein that facilitates its function. Prosthetic groups include coenzymes, which are the prosthetic groups of enzymes. The enzyme in complex I is NADH dehydrogenase and is composed of 44 separate polypeptide chains. Complex I can pump four hydrogen ions across the membrane from the matrix into the intermembrane space, and it is in this way that the hydrogen ion gradient is established and maintained between the two compartments separated by the inner mitochondrial membrane.
Q and Complex II
Complex II directly receives FADH2, which does not pass-through complex I. The compound connecting the first and second complexes to the third is ubiquinone B. The Q molecule is lipid soluble and freely moves through the hydrophobic core of the membrane. Once it is reduced (QH2), ubiquinone delivers its electrons to the next complex in the electron transport chain. Q receives the electrons derived from NADH from complex I, and the electrons derived from FADH2 from complex II. This enzyme and FADH2 form a small complex that delivers electrons directly to the electron transport chain, bypassing the first complex. Since these electrons bypass and thus do not energize the proton pump in the first complex, fewer ATP molecules are made from the FADH2 electrons. The number of ATP molecules ultimately obtained is directly proportional to the number of protons pumped across the inner mitochondrial membrane.
Complex III
The third complex is composed of cytochrome b—another Fe-S protein, a Rieske center (2Fe-2S center), and cytochrome c proteins. This complex is also called cytochrome oxidoreductase. Cytochrome proteins have a prosthetic group of heme. The heme molecule is similar to the heme in hemoglobin, but it carries electrons, not oxygen. As a result, the iron ion at its core is reduced and oxidized as it passes the electrons, fluctuating between different oxidation states: Fe++ (reduced) and Fe+++ (oxidized). The heme molecules in the cytochromes have different natures depending on how it is bound to the different proteins. Thus, giving each complex a slightly different characteristic. Complex III pumps out protons through the membrane and passes its electrons to cytochrome c for transport to complex four (Q carries pairs of electrons, whereas cytochrome c only carries one electron at a time).
Complex IV
The fourth complex is composed of cytochrome proteins c, a, and a3. This complex contains two heme groups (one in each of the two cytochromes, a, and a3) and three copper ions (a pair of CuA and one CuB in cytochrome a3). The cytochromes hold an oxygen molecule very tightly between the iron and copper ions until the oxygen is completely reduced by the gain of two electrons. The reduced oxygen then picks up two hydrogen ions from the surrounding medium to make water (H2O). The removal of the hydrogen ions from the system contributes to the ion gradient that forms the foundation for the process of chemiosmosis.
Access for free at https://openstax.org/books/biology-2e/pages/7-4-oxidative-phosphorylation
Chemiosmosis and ATP Production
In chemiosmosis, the free energy from the series of redox reactions is used to pump hydrogen ions (protons) across the mitochondrial membrane. The uneven distribution of H+ ions across the membrane establishes both concentration and electrical gradients (thus, an electrochemical gradient), owing to the hydrogen ions’ positive charge and their aggregation on one side of the membrane.
If the membrane were continuously open to simple diffusion by the hydrogen ions, the ions would tend to diffuse back across into the matrix, driven by the concentrations producing their electrochemical gradient. Recall that many ions cannot diffuse through the nonpolar regions of phospholipid membranes without the aid of ion channels. Similarly, hydrogen ions in the matrix space can only pass through the inner mitochondrial membrane by an integral membrane protein called ATP synthase (Figure 2.1.13). This complex protein acts as a tiny generator producing ATP as the hydrogen ions diffuse back into the mitochondria matrix along their concentration gradient. The turning parts of this molecular machine facilitate the addition of a phosphate to ADP, forming ATP, using the potential energy of the hydrogen ion gradient.
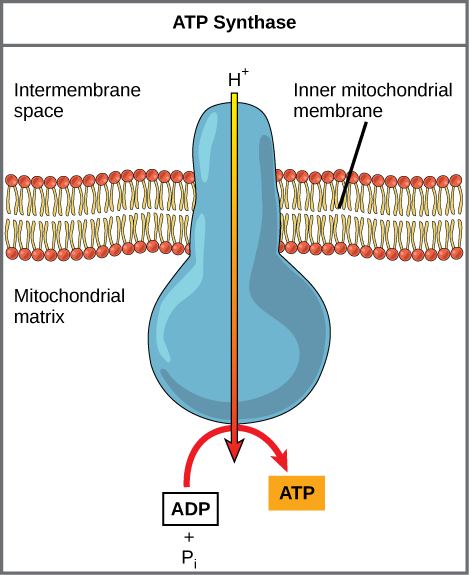
Chemiosmosis (Figure 2.1.14) is used to generate 90 percent of the ATP made during aerobic glucose catabolism; it is also the method used in the light reactions of photosynthesis to harness the energy of sunlight in the process of photophosphorylation. Recall that the production of ATP using the process of chemiosmosis in mitochondria is called oxidative phosphorylation. The overall result of these reactions is the production of ATP from the energy of the electrons removed from hydrogen atoms. These atoms were originally part of a glucose molecule. At the end of the pathway, the electrons are used to reduce an oxygen molecule to oxygen ions. The extra electrons on the oxygen attract hydrogen ions (protons) from the surrounding medium, and water is formed. Thus, oxygen is the final electron acceptor in the electron transport chain.
ATP Yield
The number of ATP molecules generated from the catabolism of glucose varies. For example, the number of hydrogen ions that the electron transport chain complexes can pump through the membrane varies between species. Another source of variance stems from the shuttle of electrons across the membranes of the mitochondria. (The NADH generated from glycolysis cannot easily enter mitochondria.) Thus, electrons are picked up on the inside of mitochondria by either NAD+ or FAD+. As you have learned earlier, these FAD+ molecules can transport fewer ions; consequently, fewer ATP molecules are generated when FAD+ acts as a carrier. NAD+ is used as the electron transporter in the liver and FAD+ acts in the brain.
Another factor that affects the yield of ATP molecules generated from glucose is the fact that intermediate compounds in these pathways are also used for other purposes. Glucose catabolism connects with the pathways that build or break down all other biochemical compounds in cells, and the result is somewhat messier than the ideal situations described thus far. For example, sugars other than glucose are fed into the glycolytic pathway for energy extraction. In addition, the five-carbon sugars that form nucleic acids are made from intermediates in glycolysis. Certain nonessential amino acids can be made from intermediates of both glycolysis and the citric acid cycle. Lipids, such as cholesterol and triglycerides, are also made from intermediates in these pathways, and both amino acids and triglycerides are broken down for energy through these pathways. Overall, in living systems, these pathways of glucose catabolism extract about 34 percent of the energy contained in glucose, with the remainder being released as heat.
Access for free at https://openstax.org/books/biology-2e/pages/7-4-oxidative-phosphorylation
Anaerobic Cellular Respiration
In aerobic respiration, the final electron acceptor is an oxygen molecule, O2. If aerobic respiration occurs, then ATP will be produced using the energy of high-energy electrons carried by NADH or FADH2 to the electron transport chain. If aerobic respiration does not occur, NADH must be re-oxidized to NAD+ for reuse as an electron carrier for the glycolytic pathway to continue. In order to accomplish this, some living systems use an organic molecule as the final electron acceptor. Processes that use an organic molecule to regenerate NAD+ from NADH are collectively referred to as fermentation. In contrast, some living systems use an inorganic molecule as a final electron acceptor. Both methods are called anaerobic cellular respiration, in which organisms convert energy for their use in the absence of oxygen.
Visit this site to see fermentation in action.
Certain prokaryotes, including some species in the domains Bacteria and Archaea, use anaerobic respiration. For example, a group of archaeans called methanogens reduces carbon dioxide to methane to oxidize NADH. These microorganisms are found in the soil and in the digestive tracts of ruminants, such as cows and sheep. Similarly, sulfate-reducing bacteria, most of which are anaerobic (Figure 2.1.15), reduce sulfate to hydrogen sulfide to regenerate NAD+ from NADH.

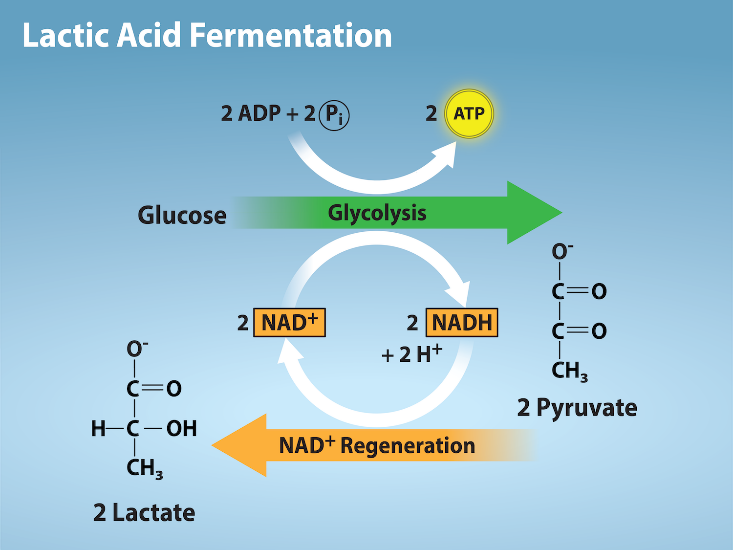
Lactic Acid Fermentation
The fermentation method used by animals and certain bacteria, such as those in yogurt, is lactic acid fermentation (Figure 2.1.16). This type of fermentation is used routinely in mammalian red blood cells, which do not have mitochondria, and in skeletal muscle that has an insufficient oxygen supply to allow aerobic respiration to continue (that is, in muscles used to the point of fatigue). In muscles, lactic acid accumulation must be removed by the blood circulation, and when the lactic acid loses hydrogen, the resulting lactate is brought to the liver for further metabolism. The chemical reactions of lactic acid fermentation are the following:
Pyruvic acid + NADH ↔ lactic acid + NAD+
The enzyme used in this reaction is lactate dehydrogenase (LDH). The reaction can proceed in either direction, but the reaction from left to right is inhibited by acidic conditions. Such lactic acid accumulation was once believed to cause muscle stiffness, fatigue, and soreness, although more recent research disputes this hypothesis. Once the lactic acid has been removed from the muscle and circulated to the liver, it can be reconverted into pyruvic acid and further catabolized for energy.
Alcohol Fermentation
Another familiar fermentation process is alcohol fermentation (Figure 2.1.17), which produces ethanol. The first chemical reaction of alcohol fermentation is the following (CO2 does not participate in the second reaction):
pyruvic acid + H+ → CO2 + acetaldehyde + NADH + H+ → ethanol + NAD+
The first reaction is catalyzed by pyruvate decarboxylase, a cytoplasmic enzyme, with a coenzyme of thiamine pyrophosphate (TPP, derived from vitamin B1—thiamine). A carboxyl group is removed from pyruvic acid, releasing carbon dioxide as a gas. The loss of carbon dioxide reduces the size of the molecule by one carbon, producing acetaldehyde. The second reaction is catalyzed by alcohol dehydrogenase to oxidize NADH to NAD+ and reduce acetaldehyde to ethanol. The fermentation of pyruvic acid by yeast produces the ethanol found in alcoholic beverages. The ethanol tolerance of yeast is variable, ranging from about 5 percent to 21 percent, depending on the yeast strain and environmental conditions.

Other Types of Fermentation
Other fermentation methods take place in bacteria. We should note that many prokaryotes are facultatively anaerobic. This means that they can switch between aerobic respiration and fermentation, depending on the availability of free oxygen. Certain prokaryotes, such as Clostridia, are obligate anaerobes. Obligate anaerobes live and grow in the absence of molecular oxygen. Oxygen is a poison to these microorganisms and kills them on exposure. We should also note that all forms of fermentation, except lactic acid fermentation, produce gas. The production of particular types of gas is used as an indicator of the fermentation of specific carbohydrates, which plays a role in the laboratory identification of the bacteria. Various methods of fermentation are used by assorted organisms to ensure an adequate supply of NAD+ for the sixth step in glycolysis. Without fermentation, this step would not occur, and ATP could not be harvested from the breakdown of glucose.
Access for free at https://openstax.org/books/biology-2e/pages/7-5-metabolism-without-oxygen
Regulatory Mechanisms
Cellular respiration must be regulated in order to provide balanced amounts of energy in the form of ATP. The cell also must generate a number of intermediate compounds that are used in the anabolism and catabolism of macromolecules. Without controls, metabolic reactions would quickly come to a standstill as the forward and backward reactions reached a state of equilibrium. Resources would be used inappropriately. A cell does not need the maximum amount of ATP that it can make all the time: At times, the cell needs to shunt some of the intermediates to pathways for amino acid, protein, glycogen, lipid, and nucleic acid production. In short, the cell needs to control its metabolism. A variety of mechanisms are used to control cellular respiration. Some type of control exists at each stage of glucose metabolism.
Some reactions are controlled by having two different enzymes—one each for the two directions of a reversible reaction. Reactions that are catalyzed by only one enzyme can go to equilibrium, stalling the reaction. In contrast, if two different enzymes (each specific for a given direction) are necessary for a reversible reaction, the opportunity to control the rate of the reaction increases, and equilibrium is not reached.
A number of enzymes involved in each of the pathways—in particular, the enzyme catalyzing the first committed reaction of the pathway—are controlled by the attachment of a molecule to an allosteric site on the protein. The molecules most commonly used in this capacity are the nucleotides ATP, ADP, AMP, NAD+, and NADH. These regulators—allosteric effectors—may increase or decrease enzyme activity, depending on the prevailing conditions. The allosteric effector alters the steric structure of the enzyme, usually affecting the configuration of the active site. This alteration of the protein’s (the enzyme’s) structure either increases or decreases its affinity for its substrate, with the effect of increasing or decreasing the rate of the reaction. The attachment signals to the enzyme. This binding can increase or decrease the enzyme’s activity, providing a feedback mechanism. This feedback type of control is effective as long as the chemical affecting it is attached to the enzyme. Once the overall concentration of the chemical decreases, it will diffuse away from the protein, and the control is relaxed.
Visit this site to see an animation of the electron transport chain and ATP synthesis.
Evolution Connection: Cellular respiration and Oxidative Phosphorylation and Photosynthesis
The process of photosynthesis and cellular metabolism consists of several very complex pathways. It is generally thought that the first cells arose in an aqueous environment – a “soup” of nutrients – possibly on the surface of some porous clays, perhaps in the warm marine environment.
An early form of photosynthesis developed that harnessed the sun’s energy using water as the source of hydrogen atoms, but this pathway did not produce free oxygen – anoxygenic photosynthesis. Another type of anoxygenic photosynthesis did not produce free oxygen because it did not use water as the source of hydrogen ions; instead, it used materials such as hydrogen sulfide and consequently produced sulfur. It is thought that glycolysis developed at this time and could take advantage of the simple sugars being produced but that these reactions were unable to fully extract the energy stored in the carbohydrates. The development of glycolysis probably predates the evolution of photosynthesis, as it was well suited to extract energy from materials spontaneously accumulating in the “primeval soup”. A later form of photosynthesis used water as a source of electrons and hydrogen and generated free oxygen. Over time, the atmosphere accumulated oxygen. This released oxygen caused metals to undergo oxidation in the oceans and created a “rust” layer in the sediment, permitting the dating of the rise of the first oxygen-producing phototrophs. Living things adapted to exploit this new atmosphere that allowed aerobic respiration as we know it to evolve. When the process of oxygenic photosynthesis evolved cells were finally able to use the oxygen expelled by photosynthesis to extract considerably more energy from the sugar molecules using the citric acid cycle and oxidative phosphorylation.
Access for free at Access for free at https://openstax.org/books/biology-2e/pages/30-2-stems and https://openstax.org/books/biology-2e/pages/7-7-regulation-of-cellular-respiration
Glossary
acetyl CoA- combination of an acetyl group derived from pyruvic acid and coenzyme A, which is made from pantothenic acid (a B-group vitamin)
ATP synthase - (also F1F0 ATP synthase) membrane-embedded protein complex that adds a phosphate to ADP with energy from protons diffusing through it
Chemiosmosis - process in which there is a production of adenosine triphosphate (ATP) in cellular metabolism by the involvement of a proton gradient across a membrane
dephosphorylation - removal of a phosphate group from a molecule
Isomerase - enzyme that converts a molecule into its isomer
phosphorylation - addition of a high-energy phosphate to a compound, usually a metabolic intermediate, a protein, or ADP
prosthetic group - (also prosthetic cofactor) molecule bound to a protein that facilitates the function of the protein
redox reaction - chemical reaction that consists of the coupling of an oxidation reaction and a reduction reaction
substrate-level phosphorylation - production of ATP from ADP using the excess energy from a chemical reaction and a phosphate group from a reactant
Ubiquinone - soluble electron transporter in the electron transport chain that connects the first or second complex to the third
Attributions
Biology 2e By Mary Ann Clark, Matthew Douglas, Jung Choi. OpenStax is licensed under Creative Commons Attribution License v4.0
Introduction to Organismal Biology at https://sites.gatech.edu/organismalbio/ is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Botany (Ha, Morrow, and Algiers) is shared under a CC BY-NC 4.0 license and was authored, remixed, and/or curated by Melissa Ha, Maria Morrow, & Kammy Algiers.