- Author:
- Anna McCollum, ALka Sharma, Jillian Gorrell, Amanda Spangler, Madonna Kemp
- Subject:
- Agriculture, Biology
- Material Type:
- Diagram/Illustration, Textbook
- Level:
- High School
- Tags:
- License:
- Creative Commons Attribution
- Language:
- English
- Media Formats:
- Graphics/Photos
2.3 Light Dependent Reaction
2.4 Light Independent Reaction
2.5 C4 & CAM Pathways as Means of Reducing Photorespiration
2.6 How Environmental Factors Affect Photosynthesis
2.7 The Energy Cycle
2_Photosynthesis
Photosynthesis
Overview
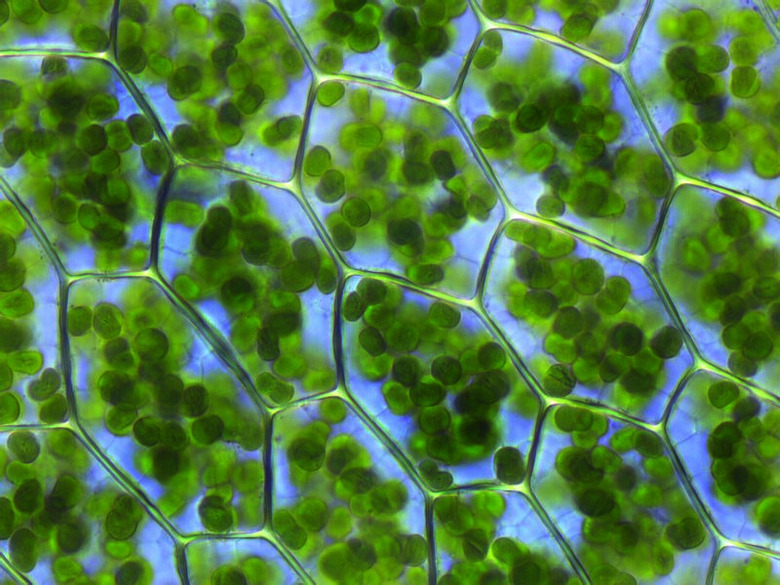
Plastids; Kristian Peters -- Fabelfroh, CC BY-SA 3.0 <http://creativecommons.org/licenses/by-sa/3.0/>, via Wikimedia Commons
Bear, Robert; Rintoul, David; Snyder, Bruce; Smith-Caldas, Martha; Herren, Christopher; and Horne, Eva, "Principles of Biology" (2016). Open Access Textbooks. 1.
https://newprairiepress.org/textbooks/1
Did you have an idea for improving this content? We’d love your input.
Introduction
Learning Objectives
- Explain the structure of chloroplast.
- Describe the steps in the light-dependent reaction of photosynthesis.
- Describe the steps in the light-independent reaction of photosynthesis.
- Differentiate between C3, C4, and CAM photosynthesis.
- List the effects of high-intensity and low-intensity light on photosynthesis.
- Explain how plants adapt to changes in solar intensity.
- List the drought and flood adaptations.
- Explain how temperature, CO2 concentration, and air movement affect photosynthesis.
Key Terms
3-phosphoglycerate - the first compound formed after CO2 assimilation by Rubisco
Accessary pigment - light-absorbing pigments other than chlorophyll a
C3 plants - plants in which the first product of CO2 fixation by Rubisco is a 3 carbon compound
C4 plants - plants in which the first product of CO2 fixation is a 4 carbon compound
CAM plants - plants that temporally separate light-dependent and light-independent reactions of photosynthesis
Calvin cycle - light-independent reactions of photosynthesis that convert carbon dioxide from the atmosphere into carbohydrates using the energy and reducing power of ATP and NADPH
Carbon fixation - a process of converting inorganic CO2 gas into organic compounds
Carotenoid - photosynthetic pigment (yellow-orange-red) that functions to dispose of excess energy
Chlorophyll - Two kinds of chlorophyll, a that absorb violet-blue and red light and consequently have a bluish-green color; the only pigment molecule that performs the photochemistry by getting excited and losing an electron to the electron transport chain; chlorophyll b is an accessory pigment that absorbs blue and red-orange light and consequently has a yellowish-green tint.
Chloroplast - organelle in which photosynthesis takes place
Cyclic photophosphorylation - ATP production by cyclic movement of electron through photosystem I
Drought avoidance - adaptations in a plant that allow it to avoid drought conditions
Drought tolerance - adaptations in a plant that allow it to survive under drought conditions
Light-dependent reaction - the first stage of photosynthesis where certain wavelengths of the visible light are absorbed to form two energy-carrying molecules (ATP and NADPH)
Light independent reaction - the second stage of photosynthesis, through which carbon dioxide is used to build carbohydrate molecules using energy from ATP and NADPH
Light intensity - number of photons falling on a unit area of the leaf surface in unit time
NADPH - the high-energy molecule
Non-cyclic photophosphorylation - ATP and NADPH production by the movement of electrons through photosystem I and photosystem II
Photolysis - the splitting of a water molecule in presence of sunlight
Photosynthesis - the process by which autotrophs use sunlight, water, and CO2 to produce sugars
Photosystem I - integral pigment and protein complex in thylakoid membranes that uses light energy to transport electrons from plastocyanin to NADP+ (which becomes reduced to NADPH in the process)
Photosystem II - integral protein and pigment complex in thylakoid membranes that transports electrons from water to the electron transport chain; oxygen is a product of PSII
Pigment - a molecule that is capable of absorbing certain wavelengths of light and reflecting others (which accounts for its color)
secondary Pigment - same as an accessory pigment
The metabolic processes in all organisms—from bacteria to humans—require energy. To get this energy, many organisms access stored energy by eating, that is, by ingesting other organisms. But where does the stored energy in food originate? All of this energy can be traced back to photosynthesis.
Photosynthesis is essential to all life on earth; both plants and animals depend on it. It is the only biological process that can capture the energy that originates from sunlight and converts it into chemical compounds (carbohydrates) that every organism uses to power its metabolism. It is also a source of oxygen necessary for many living organisms. In brief, the energy of sunlight is “captured” to energize electrons, whose energy is then stored in the covalent bonds of sugar molecules. How long-lasting and stable are those covalent bonds? The energy extracted today by the burning of coal and petroleum products represents sunlight energy captured and stored by photosynthesis 350 to 200 million years ago during the Carboniferous Period.
Plants, algae, and a group of bacteria called cyanobacteria are the only organisms capable of performing photosynthesis (figure 2.2.2). Because they use light to manufacture their own food, they are called photoautotrophs (literally, “self-feeders using light”). Other organisms—such as animals, fungi, and most other bacteria—are termed heterotrophs (“other feeders”), because they must rely on the sugars produced by photosynthetic organisms for their energy needs. A third very interesting group of bacteria synthesize sugars not by using sunlight’s energy but by extracting energy from inorganic chemical compounds. For this reason, they are referred to as chemoautotrophs.

The importance of photosynthesis is not just that it can capture sunlight’s energy. In contrast, photosynthesis is vital because it evolved as a way to store the energy from solar radiation (the “photo-” part) to energy in the carbon-carbon bonds of carbohydrate molecules (the “-synthesis” part). Those carbohydrates are the energy source that heterotrophs use to power the synthesis of ATP via respiration. Therefore, photosynthesis powers 99 percent of Earth’s ecosystems. When a top predator, such as a wolf, preys on a deer, the wolf is at the end of an energy path that went from nuclear reactions on the surface of the sun, to visible light, to photosynthesis, to vegetation, to deer, and finally to the wolf.
Photosynthesis is a multi-step process that requires specific wavelengths of visible sunlight, carbon dioxide (which is low in energy), and water as substrates (figure 2.2.3). After the process is complete, it releases oxygen and produces glyceraldehyde-3-phosphate (G3P), as well as simple carbohydrate molecules (high in energy) that can then be converted into glucose, sucrose, or any of dozens of other sugar molecules. These sugar molecules contain energy and the energized carbon that all living things need to survive.

The following is the chemical equation for photosynthesis (figure 2.2.4):
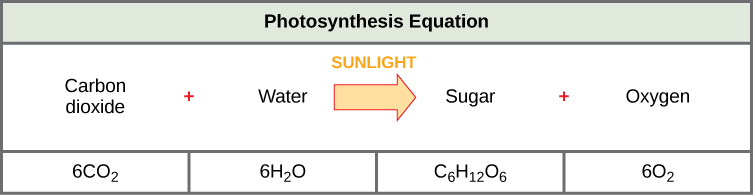
Although the equation in Figure 2.2.4. looks simple, the many steps that take place during photosynthesis are quite complex. Before learning the details of how photoautotrophs turn sunlight into food, it is important to become familiar with the structures involved.
Access for free at https://openstax.org/books/biology-2e/pages/8-1-overview-of-photosynthesis
Chloroplast
In plants, photosynthesis generally takes place in leaves, which consist of several layers of cells. The process of photosynthesis occurs in a middle layer called the mesophyll. The gas exchange of carbon dioxide and oxygen occurs through small, regulated openings called stomata (singular: stoma), which also play roles in the regulation of gas exchange and water balance. The stomata are typically located on the underside of the leaf, which helps to minimize water loss due to high temperatures on the upper surface of the leaf. Each stoma is flanked by guard cells that regulate the opening and closing of the stomata by swelling or shrinking in response to osmotic changes.
In all autotrophic eukaryotes, photosynthesis takes place inside an organelle called a chloroplast. For plants, chloroplast-containing cells exist mostly in the mesophyll. Chloroplasts have a double membrane envelope (composed of an outer membrane and an inner membrane) and are ancestrally derived from ancient free-living cyanobacteria. Within the chloroplast are stacked, disc-shaped structures called thylakoids. Embedded in the thylakoid membrane is chlorophyll, a pigment (a molecule that absorbs light) responsible for the initial interaction between light and plant material, and numerous proteins that make up the electron transport chain. The thylakoid membrane encloses an internal space called the thylakoid lumen. As shown in Figure 2.2.5, a stack of thylakoids is called a granum, and the liquid-filled space surrounding the granum is called stroma or “bed” (not to be confused with stoma or “mouth,” an opening on the leaf epidermis).
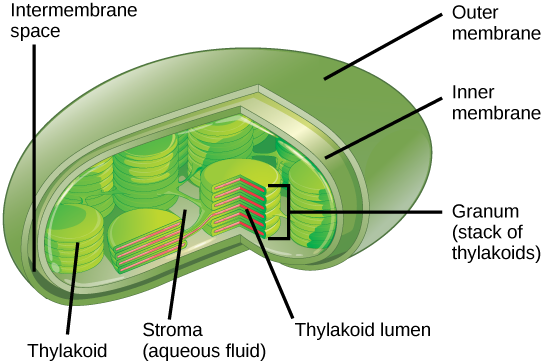
The Two Parts of Photosynthesis
Photosynthesis takes place in two sequential stages: light dependent reactions and light independent reactions. In light-dependent reactions, energy from sunlight is absorbed by chlorophyll and that energy is converted into stored chemical energy. In light-independent reactions, the chemical energy harvested during the light-dependent reactions drives the assembly of sugar molecules from carbon dioxide. Therefore, although the light-independent reactions do not use light as a reactant, they require the products of the light-dependent reactions to function. In addition, however, several enzymes of the light-independent reactions are activated by light. The light-dependent reactions utilize certain molecules to temporarily store the energy: these are referred to as energy carriers. The energy carriers that move energy from light-dependent reactions to light-independent reactions can be thought of as “full” because they are rich in energy. After the energy is released, the “empty” energy carriers return to the light-dependent reaction to obtain more energy. Figure 2.2.6 illustrates the components inside the chloroplast where light-dependent and light-independent reactions take place.
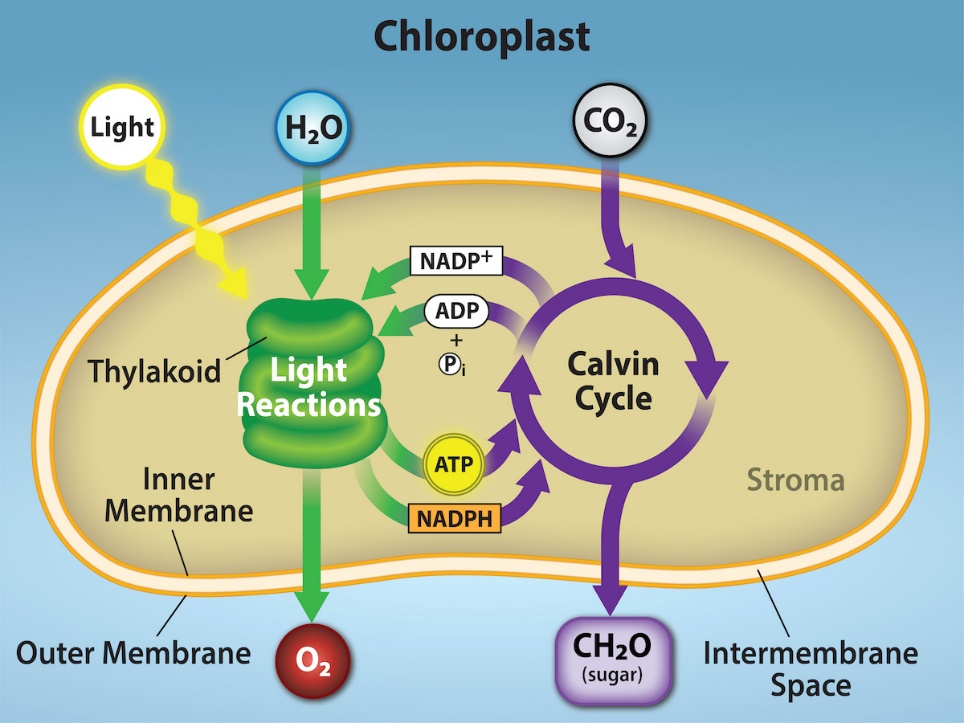
Click the link to learn more about photosynthesis.
Access for free at https://openstax.org/books/biology-2e/pages/8-1-overview-of-photosynthesis
Light Dependent Reaction
How can light energy be used to make food? When a person turns on a lamp, electrical energy becomes light energy. Like all other forms of kinetic energy, light can travel, change its form, and be harnessed to do work. In the case of photosynthesis, light energy is converted into chemical energy, which photoautotrophs use to build basic carbohydrate molecules (Figure 2.2.7). However, autotrophs only use a few specific wavelengths of sunlight.

What Is Light Energy?
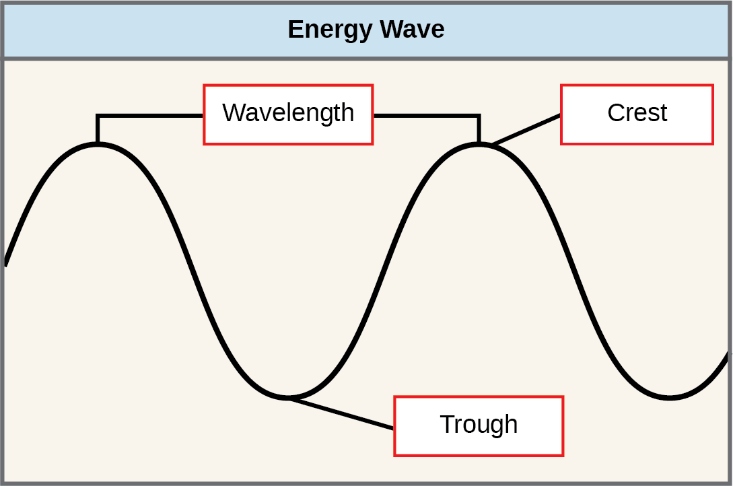
The sun emits an enormous amount of electromagnetic radiation (solar energy in a spectrum from very short gamma rays to very long radio waves). How solar energy travels is described as waves. Scientists can determine the amount of energy of a wave by measuring its wavelength (represented by the Greek symbol lambda λ)—the distance between consecutive crest points of a wave (crest to crest or from trough to trough (figure 2.2.8). The frequency of a wavelength (represented by the Greek symbol nu n) is the number of crests or troughs passing a fixed point in unit time. Thus, for any electromagnetic wave, wavelength times frequency is equal to the speed of light (represented as C, equal to 3.0 X 108 m/s).
C = λn
Shorter wavelengths have more energy than longer wavelengths. The longer the wavelength, the less energy it carries. This may seem illogical but think of it in terms of a piece of moving heavy rope. It takes little effort by a person to move a rope in long, wide waves. To make a rope move in short, tight waves, a person would need to apply significantly more energy.
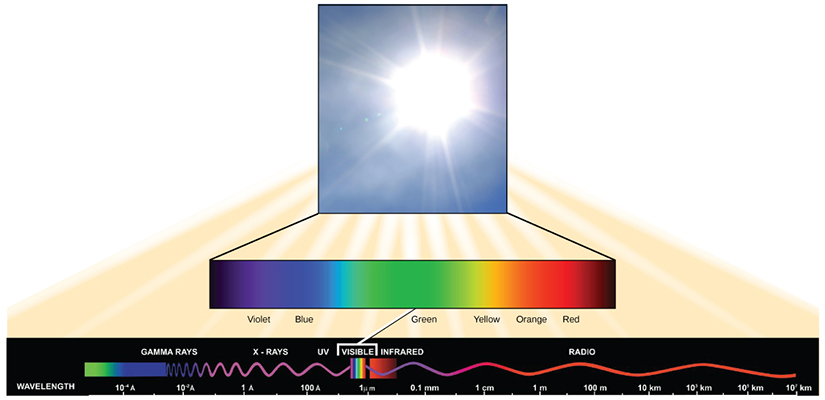
Visible light constitutes only one of many types of electromagnetic radiation emitted from the sun and other stars. Scientists differentiate the various types of radiant energy from the sun within the electromagnetic spectrum. The electromagnetic spectrum is the range of all possible frequencies of radiation (Figure 2.2.9). The difference between wavelengths relates to the amount of energy carried by them.
Electromagnetic radiations also exist as particles, called photons. All photons carry a definite amount of energy called quantum based on the wavelength. Photons are like packets of energy of an electromagnetic wave. Thus, frequency times Planck’s constant gives us the value of energy a photon is carrying.
E = hn
Where, h represents Planck’s constant (6.626 X 10-34 J s), n represents the frequency of the light.
You can visualize photons as packet of sauces that you get at any fast-food restaurant, the spiciness depends on the kind of sauces that you picked (mild, medium, or hot).
Each type of electromagnetic radiation travels at a particular wavelength. The electromagnetic spectrum (Figure 2.2.9) shows several types of electromagnetic radiation originating from the sun, including X-rays and ultraviolet (UV) rays. The higher-energy waves can penetrate tissues and damage cells and DNA, which explains why both X-rays and UV rays can be harmful to all living organisms.
Absorption of Light
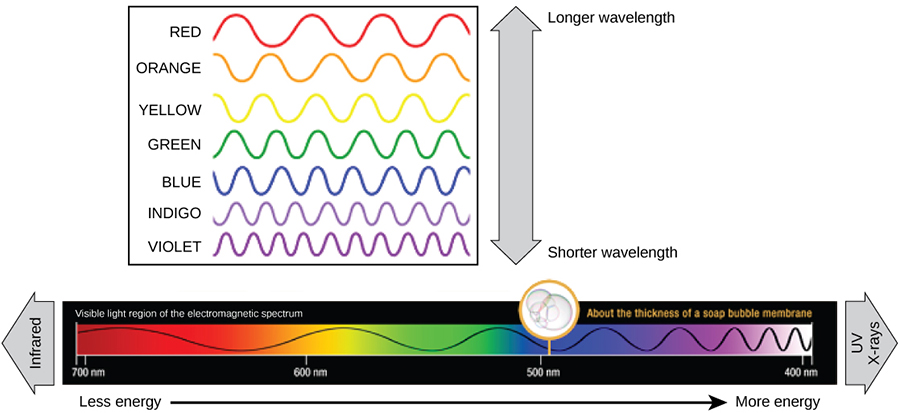
Light energy initiates the process of photosynthesis when pigments absorb specific wavelengths of visible light. Organic pigments, of the chloroplast thylakoid, have a narrow range of energy levels that they can absorb. Energy levels lower than those represented by red light are insufficient to raise an orbital electron to an excited (quantum) state. Energy levels higher than those in blue light will physically tear the molecules apart, in a process called bleaching. Our retinal pigments can only “see” (absorb) wavelengths between 700 nm and 400 nm of light, a spectrum that is therefore called visible light. For the same reasons, pigment molecules in plants, absorb only light in the wavelength range of 700 nm to 400 nm; plant physiologists refer to this range for plants as photosynthetically active radiation (Figure 2.2.10).
The visible light is seen by humans as white light exists in a rainbow of colors. Certain objects, such as a prism or a drop of water, disperse white light to reveal the colors to the human eye. The visible light portion of the electromagnetic spectrum shows the rainbow of colors, with violet and blue having shorter wavelengths, and therefore higher energy. At the other end of the spectrum toward red, the wavelengths are longer and have lower energy (Figure 2.2.11).
Understanding Pigments
Different kinds of pigments exist, and each absorbs only specific wavelengths (colors) of visible light. Pigments reflect or transmit the wavelengths they cannot absorb, making them appear a mixture of the reflected or transmitted light colors.
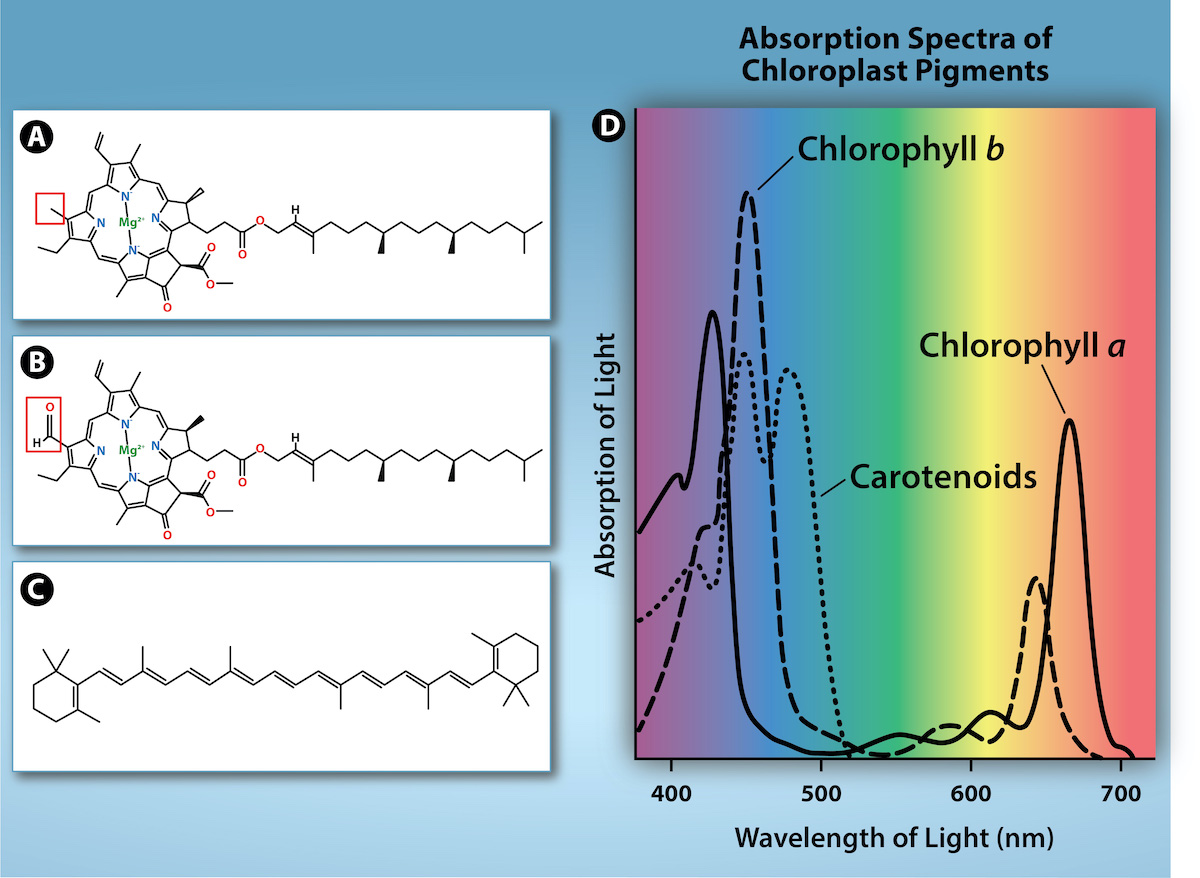
Chlorophylls and carotenoids are the two major classes of photosynthetic pigments found in plants and algae; each class has multiple types of pigment molecules. There are five major chlorophylls: a, b, c, and d, as well as a related molecule found in prokaryotes called bacteriochlorophyll. Chlorophyll a and chlorophyll b are found in the chloroplast of higher plants. With dozens of different forms, carotenoids are a much larger group of pigments. The carotenoids found in fruit—such as the red of tomato (lycopene), the yellow of corn seeds (zeaxanthin), or the orange of an orange peel (β-carotene)—are used as advertisements to attract seed dispersers. In photosynthesis, carotenoids function as photosynthetic pigments that are very efficient molecules for the disposal of excess energy. When a leaf is exposed to full sun, the light-dependent reactions are required to process an enormous amount of energy; if that energy is not handled properly, it can do significant damage. Therefore, many carotenoids reside in the thylakoid membrane, absorb excess energy, and safely dissipate that energy as heat.
Each type of pigment can be identified by the specific pattern of wavelengths it absorbs from visible light: This is termed the absorption spectrum. The graph in Figure 2.2.12 shows the absorption spectra for chlorophyll a, chlorophyll b, and a type of carotenoid pigment called β-carotene (which absorbs blue and green light). Notice how each pigment has a distinct set of peaks and troughs, revealing a highly specific pattern of absorption. Chlorophyll a absorbs wavelengths from either end of the visible spectrum (blue and red), but not green. Because green is reflected or transmitted, chlorophyll appears green. Carotenoids absorb in the short-wavelength blue region and reflect the longer yellow, red, and orange wavelengths.
Many photosynthetic organisms have a mixture of pigments, and by using these pigments, the organism can absorb energy from a wider range of wavelengths. Not all photosynthetic organisms have full access to sunlight. Some organisms grow underwater where light intensity and quality decrease and change with depth. Other organisms grow in competition for light. Plants on the rainforest floor must be able to absorb any bit of light that comes through; this is because the taller trees absorb most of the sunlight and scatter the remaining solar radiation (Figure 2.2.13).

When studying a photosynthetic organism, scientists can determine the types of pigments present by generating absorption spectra. An instrument called a spectrophotometer can differentiate which wavelengths of light a substance can absorb. A spectrophotometer measure transmitted light and compute from it the absorption. By extracting pigments from leaves and placing these samples into a spectrophotometer, scientists can identify which wavelengths of light an organism can absorb. Additional methods for the identification of plant pigments include various types of chromatography that separate the pigments by their relative affinities to solid and mobile phases.
How Light-Dependent Reactions Work
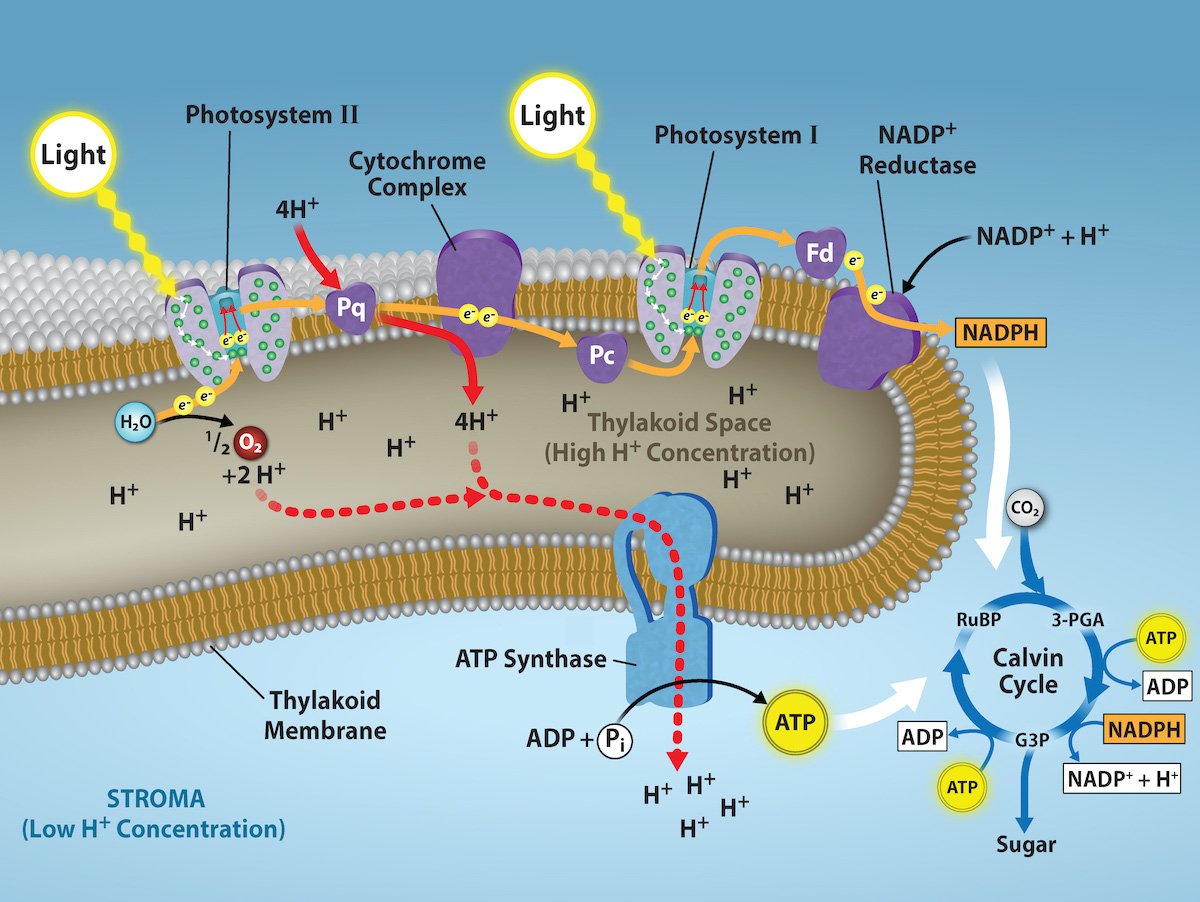
The overall function of light-dependent reactions is to convert solar energy into chemical energy in the form of NADPH and ATP. This chemical energy supports light-independent reactions and fuels the assembly of sugar molecules. The light-dependent reactions are depicted in Figure 2.2.14. Protein complexes and pigment molecules work together to produce NADPH and ATP. The numbering of the photosystems is derived from the order in which they were discovered, not in the order of the transfer of electrons.
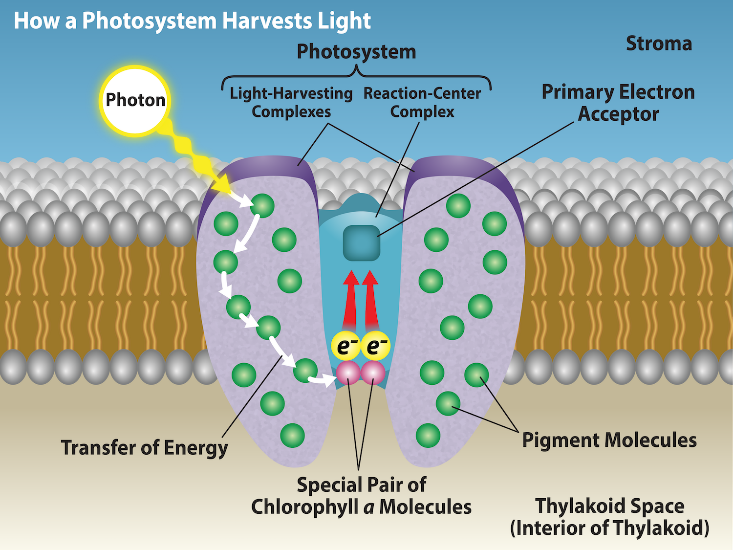
The actual step that converts light energy into chemical energy takes place in a multiprotein complex called a photosystem, two types of which are found embedded in the thylakoid membrane: photosystem II (PSII) and photosystem I (PSI) (Figure 2.2.15). The two complexes differ on the basis of what they oxidize (that is, the source of the low-energy electron supply) and what they reduce (the place to which they deliver their energized electrons).
Both photosystems have the same basic structure; a number of antenna proteins to which the chlorophyll molecules are bound surrounding the reaction center where the photochemistry takes place. Each photosystem is serviced by the light-harvesting complex, which passes energy from sunlight to the reaction center; it consists of multiple antenna proteins that contain a mixture of 300 to 400 chlorophyll a and b molecules, as well as other pigments like carotenoids. The absorption of a single photon or distinct quantity or “packet” of light by any of the chlorophylls pushes that molecule into an excited state. In short, light energy has now been captured by biological molecules but is not stored in any useful form yet. The energy is transferred from chlorophyll to chlorophyll until eventually (after about a millionth of a second), it is delivered to the reaction center. Up to this point, only energy has been transferred between molecules, not electrons.
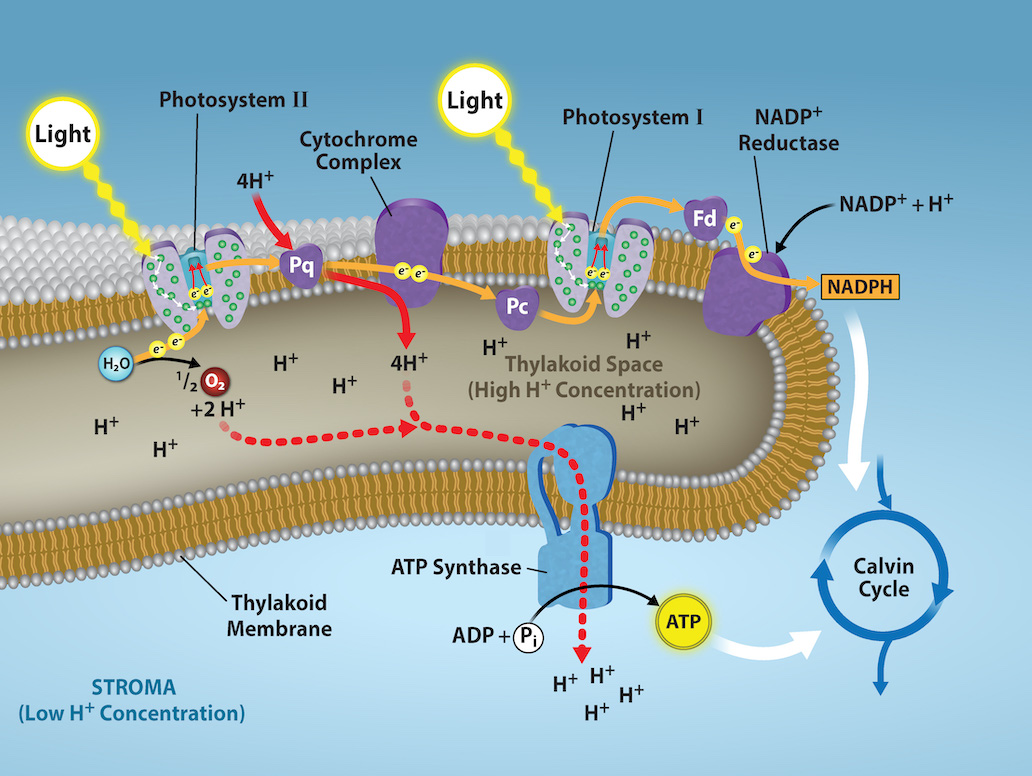
The reaction center contains a pair of chlorophyll a that have a special property. Those two chlorophylls can undergo oxidation upon excitation; they can actually give up an electron in a process that is called a photoact. It is at this step in the reaction center, during photosynthesis, that light energy is converted into an excited electron. All of the subsequent steps involve getting that electron onto the energy carrier NADPH for delivery to the Calvin cycle where the electron is deposited onto carbon for long-term storage in the form of carbohydrate. PSII and PSI are two major components of the photosynthetic electron transport chain, which also includes the cytochrome complex. The cytochrome complex, an enzyme composed of two protein complexes, transfers the electrons from the carrier molecule plastoquinone (Pq) to the protein plastocyanin (Pc), thus enabling both the transfer of protons across the thylakoid membrane and the transfer of electrons from PSII to PSI.
The reaction center of PSII (called P680) delivers its high-energy electrons, one at a time, to the primary electron acceptor, and through the electron transport chain (Pq to cytochrome complex to plastocyanin) to PSI. P680’s missing electron is replaced by extracting a low-energy electron from water; thus, water is “split” during this stage of photosynthesis, and PSII is re-reduced after every photoact. Splitting one H2O molecule releases two electrons, two hydrogen atoms, and one atom of oxygen. However, splitting two molecules is required to form one molecule of diatomic O2 gas. About 10 percent of the oxygen is used by mitochondria in the leaf to support oxidative phosphorylation. The remainder escapes to the atmosphere where it is used by aerobic organisms to support respiration.
As electrons move through the proteins that reside between PSII and PSI, they lose energy. This energy is used to move hydrogen atoms from the stromal side of the membrane to the thylakoid lumen. Those hydrogen atoms, plus the ones produced by splitting water, accumulate in the thylakoid lumen and will be used to synthesize ATP in a later step. Because the electrons have lost energy prior to their arrival at PSI, they must be re-energized by PSI, hence, another photon is absorbed by the PSI antenna. That energy is relayed to the PSI reaction center (called P700). P700 is oxidized and sends a high-energy electron to NADP+ to form NADPH. Thus, PSII captures the energy to create proton gradients to make ATP, and PSI captures the energy to reduce NADP+ into NADPH. The two photosystems partly work in concert to guarantee that the production of NADPH will roughly equal the production of ATP. Other mechanisms exist to fine-tune that ratio to exactly match the chloroplast’s constantly changing energy needs.
Generating an Energy Carrier: ATP
As in the intermembrane space of the mitochondria during cellular respiration, the buildup of hydrogen ions inside the thylakoid lumen creates a concentration gradient. The passive diffusion of hydrogen ions from high concentration (in the thylakoid lumen) to low concentration (in the stroma) is harnessed to create ATP, just as in the electron transport chain of cellular respiration. The ions build up energy because of diffusion and because they all have the same electrical charge, repelling each other.
To release this energy, hydrogen ions will rush through any opening, similar to water jetting through a hole in a dam. In the thylakoid, that opening is a passage through a specialized protein channel called the ATP synthase. The energy released by the hydrogen ion stream allows ATP synthase to attach a third phosphate group to ADP, which forms a molecule of ATP (Figure 2.2.15). The flow of hydrogen ions through ATP synthase is called chemiosmosis because the ions move from an area of high to an area of low concentration through a semi-permeable structure of the thylakoid.
Visit the site and click through the animation to view the process of photosynthesis within a leaf.
Access for free at https://openstax.org/books/biology-2e/pages/8-2-the-light-dependent-reactions-of-photosynthesis
Light Independent Reaction
After the energy from the sun is converted into chemical energy and temporarily stored in ATP and NADPH molecules, the cell has the fuel needed to build carbohydrate molecules for long-term energy storage. The products of the light-dependent reactions, ATP and NADPH—have lifespans in the range of millionths of seconds, whereas the products of the light-independent reactions (carbohydrates and other forms of reduced carbon) can survive almost indefinitely. The carbohydrate molecules made will have a backbone of carbon atoms. But where does the carbon come from? It comes from carbon dioxide—the gas that is a waste product of respiration in microbes, fungi, plants, and animals.
In plants, carbon dioxide (CO2) enters the leaves through stomata, where it diffuses over short distances through intercellular spaces until it reaches the mesophyll cells. Once in the mesophyll cells, CO2 diffuses into the stroma of the chloroplast—the site of light-independent reactions of photosynthesis. These reactions actually have several names associated with them. One of those names, the Calvin cycle, is used to honor the man who discovered it, as well as because these reactions function as a cycle. Others call it the Calvin-Benson cycle to include the name of another scientist involved in its discovery. The most outdated name is “dark reaction,” which was used because light is not directly required (figure 2.2.16). The term dark reaction can be misleading because it implies incorrectly that the reaction only occurs at night or is independent of light, which is why most scientists and instructors no longer use it.
The light-independent reactions of the Calvin cycle can be organized into three basic stages: fixation, reduction, and regeneration.
Stage 1: Fixation
In the stroma, in addition to CO2, two other components are present to initiate the light-independent reactions: an enzyme called ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO) and three molecules of ribulose bisphosphate (RuBP), as shown in Figure 2.2.17. RuBP has five atoms of carbon, flanked by two phosphates.
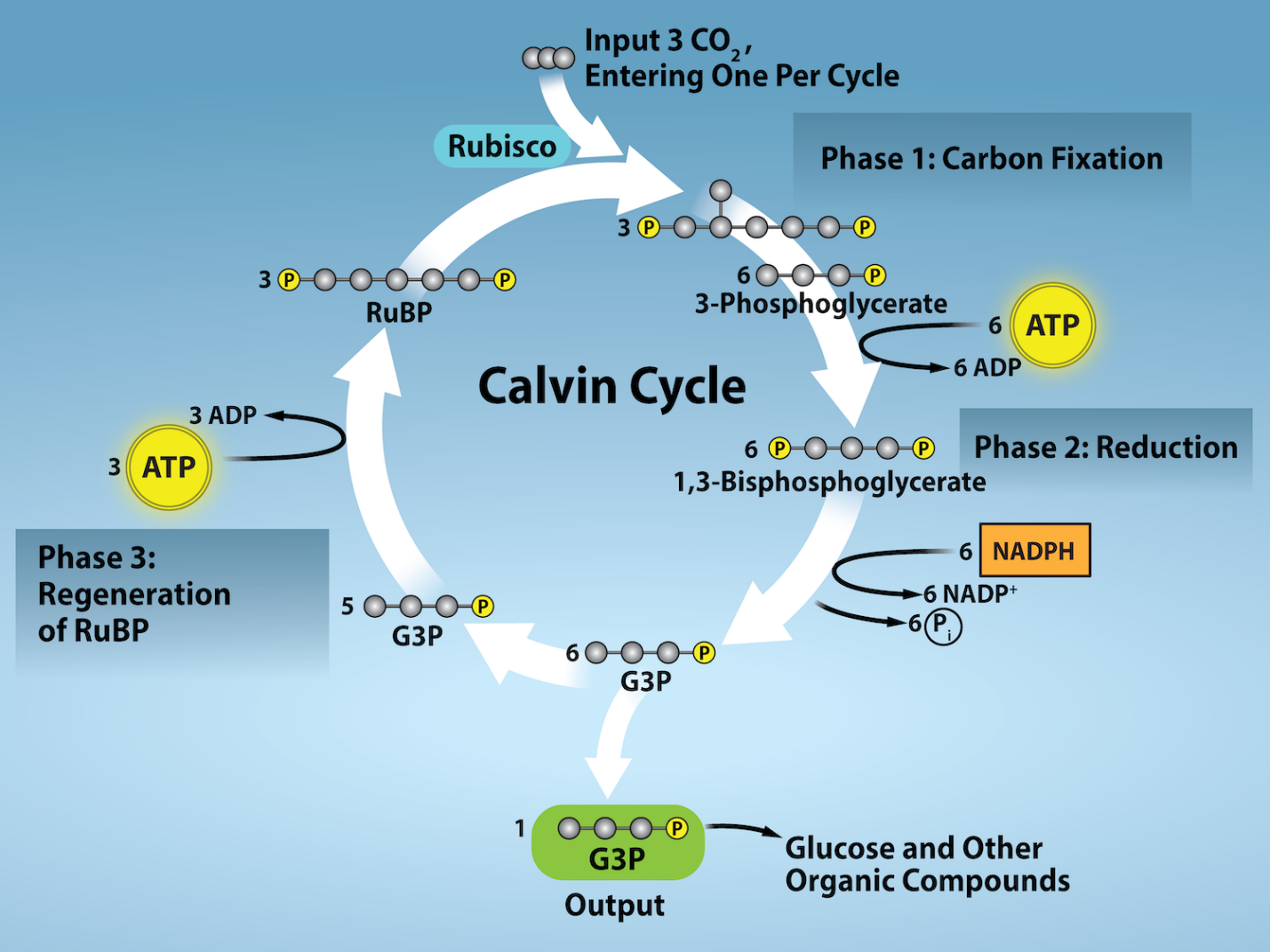
RuBisCO catalyzes a reaction between CO2 and RuBP. Each RuBP molecule combines with one CO2 molecule producing one molecule of 1, 3-bisphosphoglycerate. This molecule splits into two molecules of 3-phosphoglyceric acid (3-PGA) or 3-phosphoglycerate. PGA has three carbons and one phosphate. Each turn of the cycle involves only one RuBP and one carbon dioxide and forms two molecules of 3-PGA. The number of carbon atoms remains the same, as the atoms move to form new bonds during the reactions:
3 C atoms from 3CO2 + 15 C atoms from 3RuBP = 18 C atoms in 6 molecules of 3-PGA
This process is called carbon fixation because CO2 is “fixed” from an inorganic form into organic molecules. Since the first intermediate formed is a 3-C compound, plants that produce this compound are also called C3 plants About 85% of the plant species on the planet are C3 plants; some examples are rice, wheat, soybeans, and all trees.
Stage 2: Reduction
ATP and NADPH are used to convert the six molecules of 3-PGA into six molecules of a chemical called glyceraldehyde 3-phosphate (G3P). That is a reduction reaction because it involves the gain of electrons by 3-PGA. (Recall that a reduction is the gain of an electron by an atom or molecule.) Six molecules of both ATP and NADPH are used. For ATP, energy is released with the loss of the terminal phosphate atom, converting it into ADP; for NADPH, both energy and a hydrogen atom are lost, converting it into NADP+. Both of these molecules return to the nearby light-dependent reactions to be reused and re-energized.
Stage 3: Regeneration
Interestingly, at this point, only one of the G3P molecules leaves the Calvin cycle and is sent to the cytoplasm to contribute to the formation of other compounds needed by the plant. Because the G3P exported from the chloroplast has three carbon atoms, it takes three “turns” of the Calvin cycle to fix enough net carbon to export one G3P. But each turn makes two G3Ps, thus three turns make six G3Ps. One is exported while the remaining five G3P molecules remain in the cycle and are used to regenerate RuBP, which enables the system to prepare for more CO2 to be fixed. Three more molecules of ATP are used in these regeneration reactions.
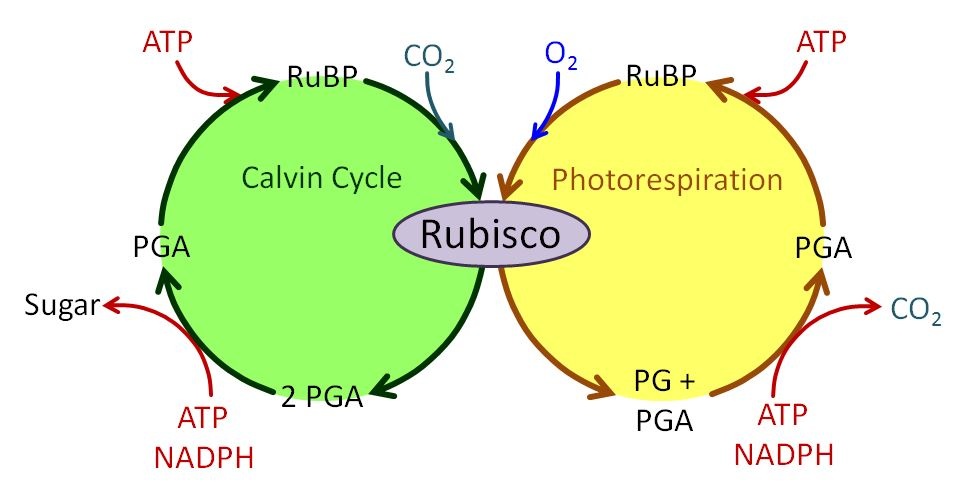
The process of photosynthesis has a theoretical efficiency of 30% (i.e., the maximum amount of chemical energy output would be only 30% of the solar energy input), but the efficiency is much lower in reality. It is only about 3% on cloudy days. Why is so much solar energy lost? There are a number of factors contributing to this energy loss, and one metabolic pathway that contributes to this low efficiency is photorespiration.
During photorespiration, the key photosynthetic enzyme Rubisco (ribulose-1,5-bisphosphate carboxylase oxygenase) uses O2 as a substrate instead of CO2. This process uses up a considerable amount of energy without making sugars (Figure 2.2.18). When a plant has its stomata open (when CO2 is diffusing in while O2 and water are diffusing out), photorespiration is minimized because Rubisco has a higher affinity for CO2 than for O2 when air temperatures are below 30°C (86°F). However, when a plant closes its stomata during times of water stress and O2 from respiration builds up inside the cell, the rate of photorespiration increases because O2 is now more abundant inside the mesophyll. So, there is a tradeoff. Plants can leave the stomata open and risk drying out, or they can close the stomata, thereby reducing the uptake of CO2, and decreasing the efficiency of photosynthesis. In addition, Rubisco has a higher affinity for O2 when temperatures increase, which means that C3 plants use more energy (ATP) for photorespiration at higher temperatures.
Access for free at https://openstax.org/books/biology-2e/pages/8-3-using-light-energy-to-make-organic-molecules
C4 & CAM Pathways as Means of Reducing Photorespiration
The C4 and CAM pathways for fixing CO2 are two adaptations that improve the efficiency of photosynthesis, by ensuring that Rubisco encounters high CO2 concentrations and thus reduces photorespiration. These two photosynthetic adaptations for fixing CO2 have evolved independently several times in species that evolved from wet and dry, but typically warm climates. Why have these mechanisms evolved independently so many times? Plants that minimize photorespiration may have a significant competitive advantage because a considerable amount of energy (in the form of ATP) is lost in plants during photorespiration. In many environments, plants that use solar energy more efficiently should out-compete those which are less efficient.
C4 Pathway
Many angiosperms have developed adaptations that minimize the losses to photorespiration. They all use a supplementary method of CO2 uptake which initially forms a four-carbon molecule compared to the two three-carbon molecules that are initially formed in the C3 pathway. Hence, these plants are called C4 plants. Note that C4 plants will eventually conduct the light-independent reactions (C3 pathway), but they form a four-carbon molecule first.
C4 plants have structural changes in their leaf anatomy (Kranz anatomy) so that synthesizing the four-carbon sugar (the C4 pathway) and resuming the light-independent reactions (C3 pathways) are separated in different parts of the leaf with RuBisCO sequestered in bundle sheath cells, where the CO2 level is high and the O2 level low. After entering through the stomata, CO2 diffuses into a mesophyll cell (Figure 2.2.19; Figure 2.2.20). Being close to the leaf surface, these cells are exposed to high levels of O2, but they have no RuBisCO so cannot start photorespiration (nor the light-independent reactions).
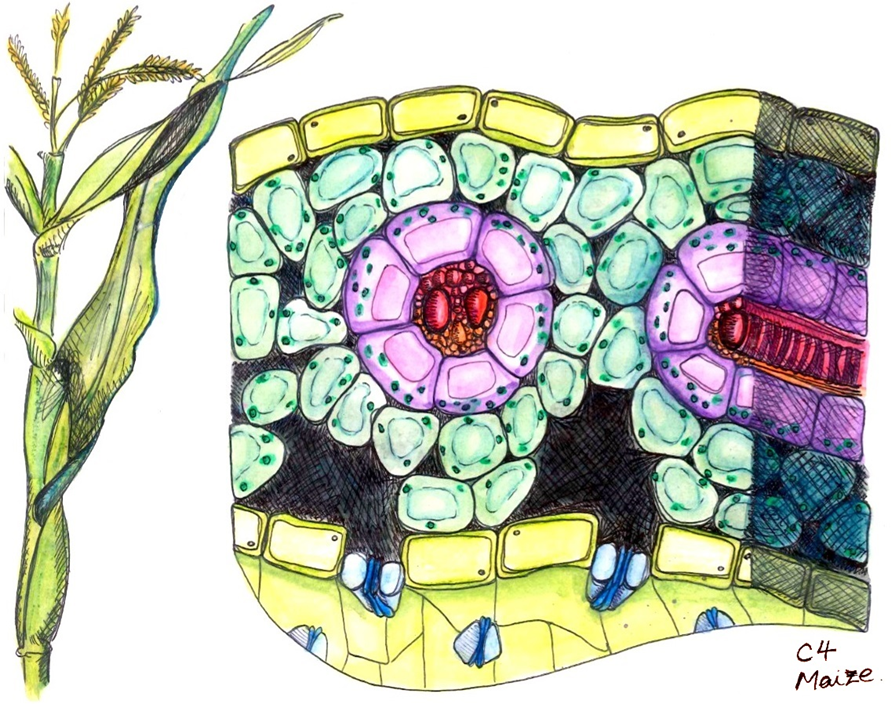
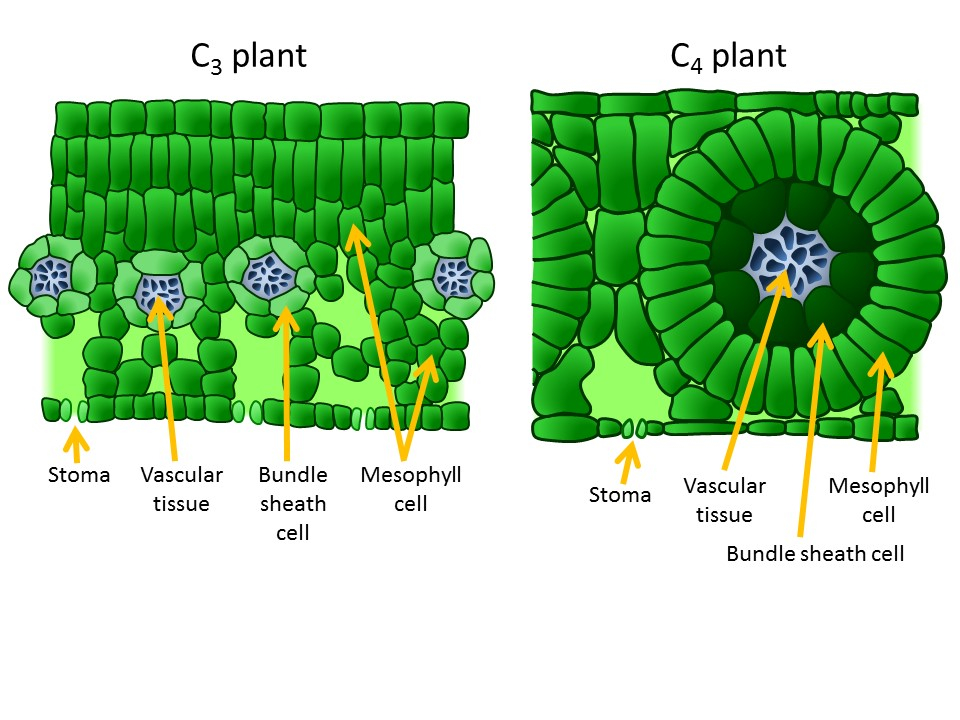
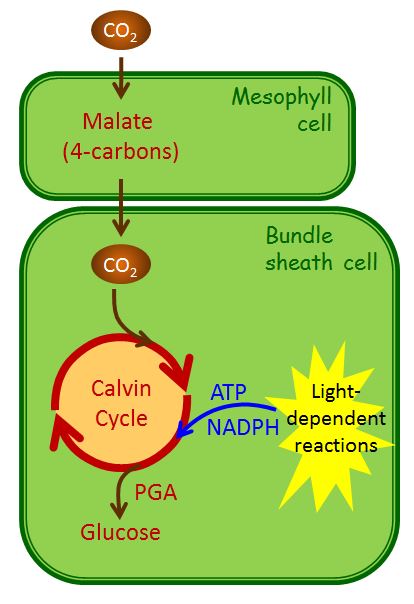
How does this work? Atmospheric CO2 is fixed in the mesophyll cells as a simple 4-carbon organic acid (malate) by an enzyme that has no affinity for O2. Malate is then transported to the bundle sheath cells. Inside the bundle sheath, malate is oxidized to a 3-C organic acid, and in the process, 1 molecule of CO2 is produced from every malate molecule (Figure 2.2.21). The CO2 is then fixed by Rubisco into sugars, via the Calvin cycle, exactly as in C3 photosynthesis. There is an additional cost of two ATPs associated with moving the three-carbon “ferry” molecule from the bundle sheath cell back to the mesophyll to pick up another molecule of atmospheric CO2. Since the spatial separation in bundle-sheath cells minimizes O2 concentrations in the locations where Rubisco is located, photorespiration is minimized (Figure 2.2.21). This arrangement of cells reduces photorespiration and increases the efficiency of photosynthesis for C4 plants. In addition, C4 plants require about half as much water as a C3 plant. The reason C4 plants require less water is that the physical shape of the stomata and leaf structure of C4 plants helps reduce water loss by developing a large CO2 concentration gradient between the outside of the leaf (400 ppm) and the mesophyll cells (10 ppm). The large CO2 concentration gradient reduces water loss via transpiration through the stomata. These C4 plants are well adapted to (and likely to be found in) habitats with high daytime temperatures and intense sunlight. Because they use the C4 pathway to prevent photorespiration, they do not have to open their stomata to the same extent as C3 plants and can thus conserve water. Some examples are crabgrass, corn (maize), sugarcane, and sorghum. Although comprising only ~3% of the angiosperms by species, C4 plants are responsible for ~25% of all the photosynthesis on land.
CAM
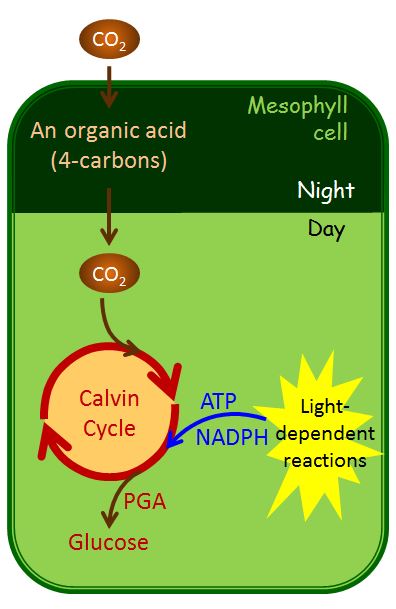
Many plants such as cacti and pineapples, which are adapted to arid environments, use different energy and water-saving pathway called crassulacean acid metabolism (CAM). This name comes from the family of plants (Crassulaceae) in which scientists first discovered the pathway. Instead of spatially separating the light-dependent reactions and the use of CO2 in the Calvin cycle, CAM plants separate these processes temporally (Figure 2.2 22). At night, CAM plants open their stomata, and an enzyme in the mesophyll cells fixes the CO2 as an organic acid; then, they store the organic acid in vacuoles until morning. During the day the light-dependent reactions supply the ATP and NADPH necessary for the Calvin cycle to function, and the CO2 is released from those organic acids in order to make sugars. Plant species using CAM photosynthesis are the most water-efficient of all; the stomata are only open at night when humidity is typically higher, and the temperatures are much cooler (which serves to lower the diffusive gradient driving water loss from leaves). The CAM pathway is primarily an adaptation to water-limited environments; the fact that this pathway also stops photorespiration is an added benefit.
Overall, C3, C4 and CAM plants all use the Calvin cycle to make sugars from CO2. Table 2.2.1, list the various ways in which plants fix CO2 and the advantages and disadvantages associated with the various mechanisms and the habitats where plants are found.
| C3 plant | C4 plant | CAM Plant |
cost | photorespiration | The cost in terms of ATP associated with fixing carbon is double. Carbon fixation is less efficient under cold conditions. | Reduced amount of fixed carbon, stomata only open at night |
benefits | Carbon fixation without using ATP | Reduced photorespiration and ability to fix Carbon under high temperatures and reduced water loss | Reduced photorespiration and reduced water loss |
Separation of light-dependent reactions and carbon fixation | None, all of these reactions occur in the same cells | Spatial, these two sets of reactions occur in different cells | Temporal, these two sets of reactions occur at different times of day |
This link explains why C4 plants can start with an alternative enzyme (PEP carboxylase) and CAM plants can alter the timing of their Calvin cycle processes to avoid/minimize O2 use and water loss.
This link leads to an animation of photosynthesis and the Calvin cycle.
How Environmental Factors Affect Photosynthesis
In the previous section, we learn how light has both wave and particle properties as well as how photons are utilized in photosynthesis. Plants grow in diverse climates and under ever-changing environmental conditions. The rate of photosynthesis also changes with changing environmental conditions. This change in photosynthetic rate directly affects crop yield. In this section, we will briefly discuss how light intensity, the concentration of CO2, high and low temperatures, water availability, and air movement affect photosynthesis.
Light Intensity
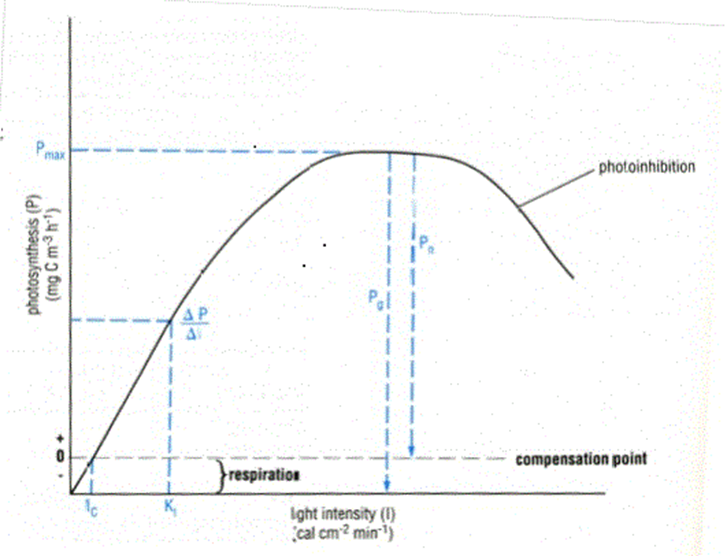
In photosynthesis, irradiance or intensity can be measured as the number of photons falling on a unit area of the leaf surface in unit time. We already know that the energy of a photon depends on the wavelength. Photosynthesis-irradiance curve (figure 2.2.23) depicts how with increasing light intensity, the rate of photosynthesis continues to increase (left portion of the graph) until it reaches a saturation point (middle portion showing flat line or plateau) and then drops (right portion of the graph). This drop in the rate of photosynthesis is due to many factors, such as CO2 concentration, temperatures change, and water conditions. When exposure to high-intensity light causes damage to photosystem II, it is called photoinhibition (Figure 2.2.24).
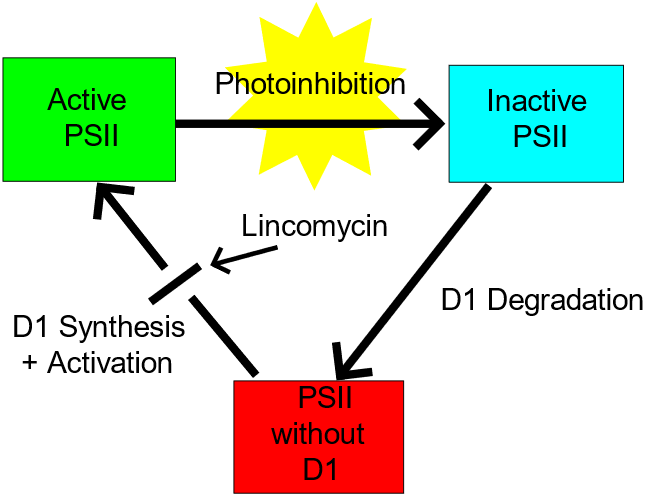
Few mechanisms that plants use to prevent photoinhibition.
- Paraheliotropism: For efficient photosynthesis, plants orient their leaves perpendicular to the incident light. To reduce the damage from photoinhibition, some plant that exhibits paraheliotropism, orient their leaves parallel to the plane of incident light.
- Chloroplast movement: cytoplasmic streaming or cyclosis has been observed in algae, mosses as well as in the leaves of higher plants. Elements of the cytoskeleton (Unit 1, lesson 1, The cell) participate in cytoplasmic streaming and move organelles such as chloroplast around the plant cell. Cytoplasmic streaming leads to changes in the location and orientation of chloroplast. Repositioning of chloroplast blocks the over-exposure to high-intensity light and prevents photoinhibition. Click this link provide to see cytoplasmic streaming in elodea leaves.
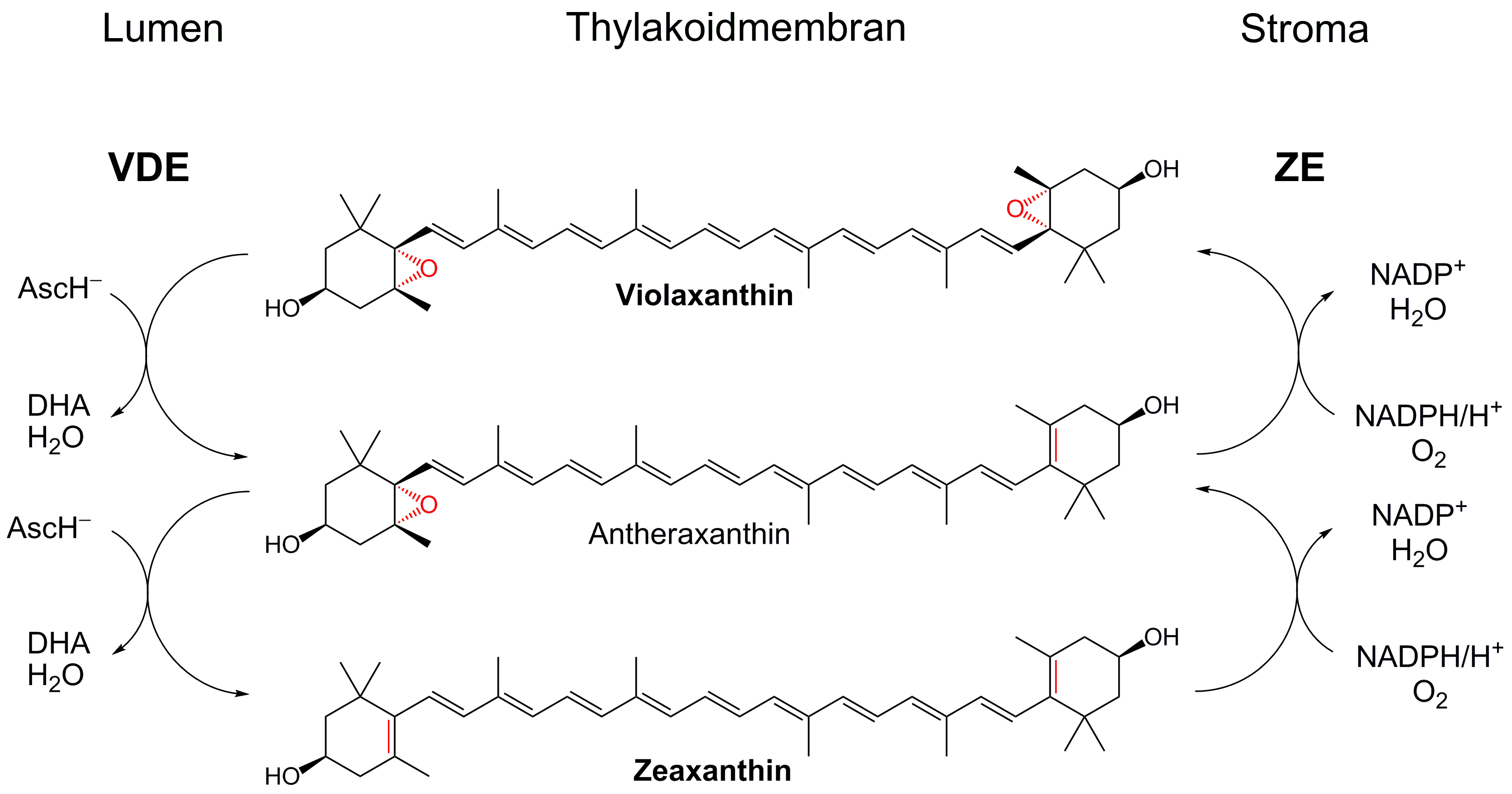
- Xanthophyll cycle: If too much light is absorbed, the pH gradient developed across the thylakoid membranes becomes greater. This is sensed by a protein, PsbS, and through subsequent conformational changes transmitted through the light-harvesting antennae, the excess light energy must be dissipated as thermal energy. Molecules called xanthophylls (synthesized from carotenes - vitamin A precursors) such as zeaxanthin are important in excess energy dissipation. These molecules appear to cause excited-state chlorophyll (singlet-like excited state dioxygen) to become de-excited (Figure 2.2.25). Without the xanthophylls, the excited state chlorophyll could deexcite by transfer of energy to ground state triplet dioxygen, promoting it to the singlet, reactive state, which through electron acquisition, could also be converted to superoxide. These reactive oxygen species (ROS) can lead to oxidative damage to proteins, lipids, and nucleic acids, alteration in gene transcription, and even programmed cell death. Carotenoids can also act as ROS scavengers. Hence both heat dissipation and inhibition of the formation of ROS (by such molecules as vitamin E) are both mechanisms of defense against excessive solar energy.
- Sun and shade leave: Plants show variation in the size and thickness of their leaves. Sun leaves are smaller and thicker than shade leaves. Thicker cuticle, more palisade parenchyma cells or layers, and more vascular tissues are observed in the sun leaves. This makes sun leaves more efficient in capturing high-intensity light while preventing water loss via stomata due to transpiration. Shade leaves are larger. This increased surface area in shade leaves increases the absorption of light (Figure 2.2.26).
CO2 concentration
The partial pressure of CO2 is identical in the atmosphere. C4 plants evolved anatomical structures called Kranz anatomy. (figure 2.2.19; unit 2 lesson 2 section 5) to concentrate CO2 while CAM plants separate CO2 fixation temporally. The rate of photosynthesis increases with increasing CO2 concentration only if stomata remain open. The opening and closing of stomata depend on the ratio of the rate of water loss by transpiration to the rate of CO2 fixation. Many factors affect this ratio. Under high CO2 concentrations, the components of light-independent reactions (RuBisCo enzyme, regeneration of Ribulose-1, 5-bisphosphate) are saturated and the rate of photosynthesis cannot be increased any further. The availability of ATP and NADPH from light-dependent reactions also limits the rate of CO2 fixation under high CO2 conditions. Photorespiration is likely to occur under low CO2 concentrations inside the leaf.
Wind speed
When winds are calm, the rate of exchange of CO2 and water vapor via stomata is slower than when there is a breeze or a gust. The rate of exchange of gases depends on the thickness of the boundary layer, a layer of stagnant air around the leaf. A breeze or a gust disrupts the boundary layer and helps in increasing the rate of gas exchange thus enhancing the rate of diffusion of CO2 into the leaves. The increased rate of CO2 diffusion supports a higher rate of photosynthesis. Similarly, on humid days, the rate of exchange of water vapor and CO2 goes down decreasing the rate of photosynthesis.
Temperature
Low and high temperatures are relative to the range of temperature where a plant is growing. The temperature range for photosynthesis is wide, 59°F to 104°F. Rate of photosynthesis increases as the temperature rises, but only up to a certain level. Plants grow in different climates and under a wide range of temperatures and are well adapted to the temperature fluctuations in the area. The probability of photorespiration increases with high temperatures. This is because, at high temperatures, RuBisCO has a lower affinity for CO2. Lower temperatures bring down the activity of multiple enzymes involved in photosynthesis as well as transport in phloem and xylem.
Water availability
As we already know, plants grow in different climates and are well adapted to the environmental conditions of those regions. Low water conditions or drought conditions disrupt the water potential of the cells and thus affect all functions including photosynthesis. Remember, CO2 exchange occurs via stomata. To do photosynthesis plants must open stomata and this can cause further aggravate the water stress. Depending on the species and climate, plants show many adaptations to avoid water loss, such as sunken stomata, thick waxy cuticle, fewer stomata, deep roots, water storage in roots or other parts of the plant, reduced growth, smaller or reduced leaf to decrease the surface area for transpiration or in some cases complete loss of leaves, for example, spines on cacti are reduced leaves. Some plants called xerophytes are naturally tolerant to drought and can survive under extreme drought conditions. Draught-tolerant plants continue to grow and function under severe drought conditions. Whereas plants that germinate, grow, and reproduce only when water is available are called drought-avoiding plants.
Flooding also reduces the rate of photosynthesis. Roots are non-photosynthetic and depend on cellular respiration to support all functions. When submerged under water for long periods roots face oxygen scarcity. Many plants develop spaces or channels within tissue to store diffused air from aerial parts of the plant. For example, water lilies develop aerenchyma (figure 2.2.27). Many plants develop adventitious roots, roots growing from stems or other parts of plants above the water level. Intertidal zones are areas of seashore where land is submerged during high tide and exposed during low tide. Mangroves are well adapted to overcome the effects of flooding due to high tides. Mangroves grow aerial roots called pneumatophores (Figure 2.2.28) that grow upwards and stick out of the water surface. Numerous lenticels cover the surface of these roots to aid in O2 absorption. Lenticels (Figure 2.2.29) are openings in the bark, made up of cells that are permeable to O2 due to a lack of suberin deposition. Some plants, such as rice and cottonwood germinate as well as grow well under flooded conditions.



The Energy Cycle
Whether the organism is a bacterium, plant, or animal, all living things access energy by breaking down carbohydrates and other carbon-rich organic molecules. But if plants make carbohydrate molecules, why would they need to break them down, especially when it has been shown that the gas organisms release as a “waste product” (CO2) acts as a substrate for the formation of more food in photosynthesis? Remember, living things need the energy to perform life functions. In addition, an organism can either make its own food or eat another organism—either way, the food still needs to be broken down. Finally, in the process of breaking down food, called cellular respiration, heterotrophs release needed energy and produce “waste” in the form of CO2 gas.
However, in nature, there is no such thing as “waste.” Every single atom of matter and energy is conserved and recycled over and over infinitely. Substances change the form or move from one type of molecule to another, but their constituent atoms never disappear (Figure 8.2.30).
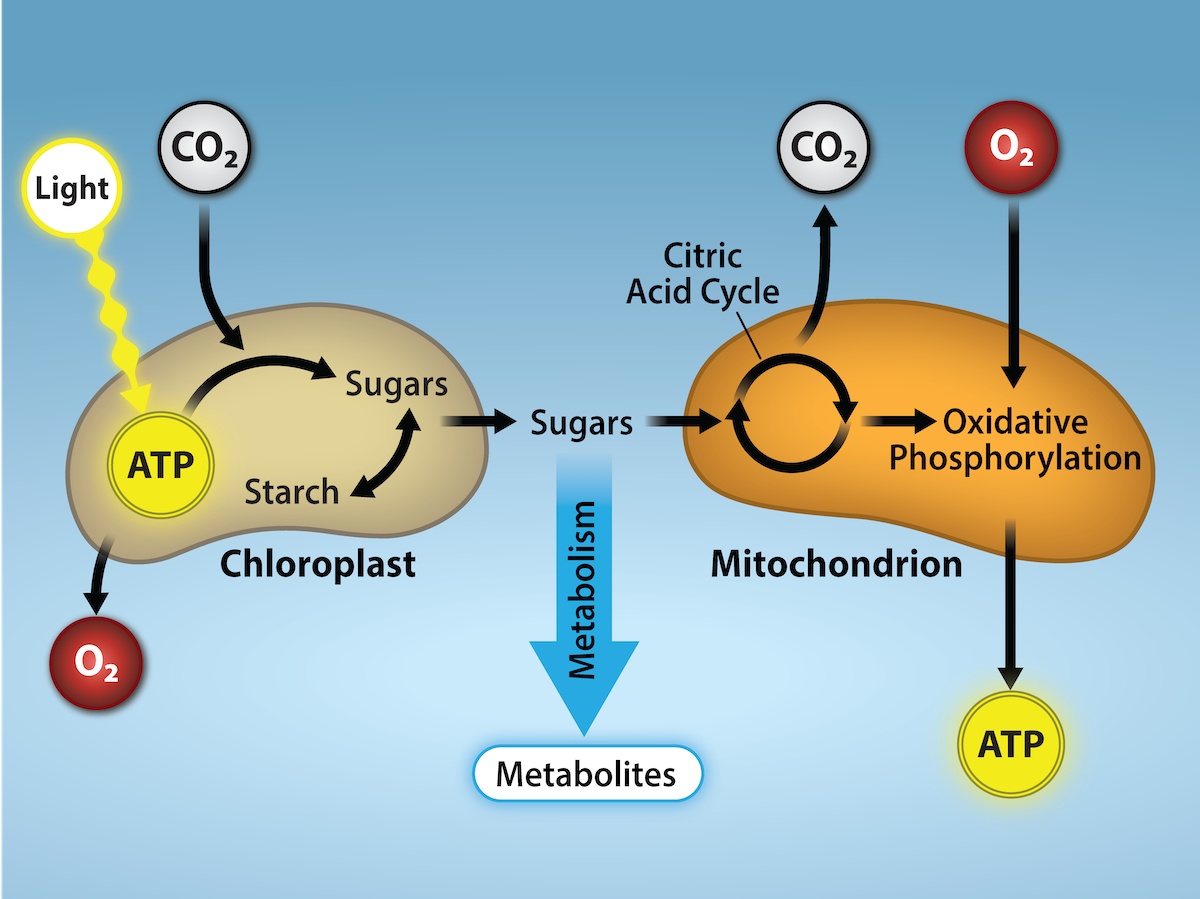
In reality, CO2 is no more a form of waste than oxygen is wasteful to photosynthesis. Both are byproducts of reactions that move on to other reactions. Photosynthesis absorbs light energy to build carbohydrates in chloroplasts, and aerobic cellular respiration releases energy by using oxygen to metabolize carbohydrates in the cytoplasm and mitochondria. Both processes use electron transport chains to capture the energy necessary to drive other reactions. These two powerhouse processes, photosynthesis and cellular respiration (Figure 2.2.31), function in biological, cyclical harmony to allow organisms to access life-sustaining energy that originates millions of miles away in a burning star humans call the sun.
Everyday Connection: Photosynthesis at the Grocery Store

Major grocery stores in the United States are organized into departments, such as dairy, meats, produce, bread, cereals, and so forth. Each aisle (figure 2.2.32) contains hundreds, if not thousands, of different products for customers to buy and consume.
Although there is a large variety, each item ultimately can be linked back to photosynthesis. Meats and dairy link because the animals were fed plant-based foods The bread, cereals, and pasta come largely from starchy grains, which are the seeds of photosynthesis in plants. What about desserts and drinks? All of these products contain sugar – sucrose in a plant product, a disaccharide, a carbohydrate molecule, which is built directly from photosynthesis. Moreover, many items are less obviously derived from plants: paper goods are generally plant products and many plastics (abundant as products and packaging) are derived from “algae” (unicellular photosynthesizing protozoans and cyanobacteria). Virtually every specie and flavoring in the spice aisle was produced by a plant as a leaf, root, bark, flower, fruit, or stem. Ultimately, photosynthesis connects to every meal and every food a person consumes.
Access for free at https://openstax.org/books/biology-2e/pages/8-3-using-light-energy-to-make-organic-molecules
Attributions
Biology 2e By Mary Ann Clark, Matthew Douglas, Jung Choi. OpenStax is licensed under Creative Commons Attribution License v4.0
Introduction to Organismal Biology at https://sites.gatech.edu/organismalbio/ is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Botany (Ha, Morrow, and Algiers) is shared under a CC BY-NC 4.0 license and was authored, remixed, and/or curated by Melissa Ha, Maria Morrow, & Kammy Algiers.
Bear, Robert; Rintoul, David; Snyder, Bruce; Smith-Caldas, Martha; Herren, Christopher; and Horne, Eva, "Principles of Biology" (2016). Open Access Textbooks. 1.
https://newprairiepress.org/textbooks/1
Glossary
absorption spectrum - range of wavelengths of electromagnetic radiation absorbed by a given substance
antenna protein - pigment molecule that directly absorbs light and transfers the energy absorbed to other pigment molecules
Calvin cycle - light-independent reactions of photosynthesis that convert carbon dioxide from the atmosphere into carbohydrates using the energy and reducing power of ATP and NADPH
carbon fixation - process of converting inorganic CO2 gas into organic compounds
carotenoid - photosynthetic pigment (yellow-orange-red) that functions to dispose of excess energy
chemoautotroph - organism that can build organic molecules using energy derived from inorganic chemicals instead of sunlight
chlorophyll a - form of chlorophyll that absorbs violet-blue and red light and consequently has a bluish-green color; the only pigment molecule that performs the photochemistry by getting excited and losing an electron to the electron transport chain
chlorophyll b - accessory pigment that absorbs blue and red-orange light and consequently has a yellowish-green tint
chloroplast - organelle in which photosynthesis takes place
cytochrome complex - group of reversibly oxidizable and reducible proteins that forms part of the electron transport chain between photosystem II and photosystem I
electromagnetic spectrum - range of all possible frequencies of radiation
electron transport chain - group of proteins between PSII and PSI that pass energized electrons and use the energy released by the electrons to move hydrogen ions against their concentration gradient into the thylakoid lumen
granum - stack of thylakoids located inside a chloroplast
heterotroph - organism that consumes organic substances or other organisms for food
light harvesting complex - complex that passes energy from sunlight to the reaction center in each photosystem; consists of multiple antenna proteins that contain a mixture of 300 to 400 chlorophyll a and b molecules, as well as other pigments like carotenoids
light-dependent reaction - first stage of photosynthesis where certain wavelengths of the visible light are absorbed to form two energy-carrying molecules (ATP and NADPH)
light-independent reaction - second stage of photosynthesis, through which carbon dioxide is used to build carbohydrate molecules using energy from ATP and NADPH
mesophyll - middle layer of chlorophyll-rich cells in a leaf
P680 - reaction center of photosystem II
P700 - reaction center of photosystem I
photoact - ejection of an electron from a reaction center using the energy of an absorbed photon
photoautotroph - organism capable of producing its own organic compounds from sunlight
photon - distinct quantity or “packet” of light energy
photosystem - group of proteins, chlorophyll, and other pigments that are used in the light-dependent reactions of photosynthesis to absorb light energy and convert it into chemical energy
photosystem I - integral pigment and protein complex in thylakoid membranes that uses light energy to transport electrons from plastocyanin to NADP+ (which becomes reduced to NADPH in the process)
photosystem II - integral protein and pigment complex in thylakoid membranes that transports electrons from water to the electron transport chain; oxygen is a product of PSII
pigment - molecule that is capable of absorbing certain wavelengths of light and reflecting others (which accounts for its color)
primary electron acceptor - pigment or other organic molecule in the reaction center that accepts an energized electron from the reaction center
reaction center - complex of chlorophyll molecules and other organic molecules that is assembled around a special pair of chlorophyll molecules and a primary electron acceptor, capable of undergoing oxidation and reduction
reduction - gain of electron(s) by an atom or molecule
spectrophotometer - instrument that can measure transmitted light and compute the absorption
stoma - opening that regulates gas exchange and water evaporation between leaves and the environment, typically situated on the underside of leaves
stroma - fluid-filled space surrounding the grana inside a chloroplast where the light-independent reactions of photosynthesis take place
thylakoid - disc-shaped, membrane-bound structure inside a chloroplast where the light-dependent reactions of photosynthesis take place; stacks of thylakoids are called grana
thylakoid lumen - aqueous space bound by a thylakoid membrane where protons accumulate during light-driven electron transport
wavelength - distance between consecutive points of equal position (two crests or two troughs) of a wave in a graphic representation; inversely proportional to the energy of the radiation