- Author:
- Anna McCollum, ALka Sharma, Jillian Gorrell, Amanda Spangler, Madonna Kemp
- Subject:
- Agriculture, Life Science
- Material Type:
- Textbook
- Level:
- High School, Community College / Lower Division
- Tags:
- License:
- Creative Commons Attribution Non-Commercial Share Alike
- Language:
- English
5.3 Major and Minor Nutrients
5.4 Nitrogen Fixation
5.5 Mycorrhizae_The Symbiotic Relationship between Fungi and Roots
5_Plant-Nutrition
Plant Nutrition
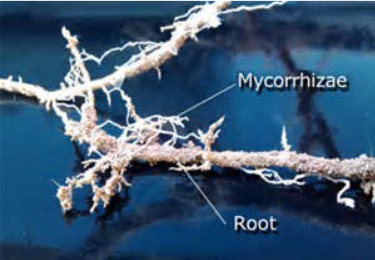
Overview
Title Image "Root with Mycorhhizae" by the United States Department of Agriculture, Natural Resources Conservation Service is in the Public Domain.
Did you have an idea for improving this content? We’d love your input.
Introduction
Learning Objectives
Distinguish between the major and minor plant nutrients and deficiency symptoms.
Identify the major and minor plant nutrients.
List the most common symptoms of nutrient deficiency in plants.
Explain the process of nitrogen fixation by bacteria.
Describe phosphorus absorption.
Explain the process of primary and secondary ecological succession.
Key Terms
biomolecule - any organic compound that is produced by living organisms
essential element - elements that are directly involved in plant nutrition, perform a function that no other element can, and are necessary for a plant to complete its life cycle
macronutrient - nutrient that is required in large amounts for plant growth
micronutrient - nutrient required in small amounts; also called trace element
mycorrhizae - a symbiotic association between a plant and a fungus
nitrogenase - enzyme that is responsible for the reduction of atmospheric nitrogen to ammonia
nutrient deficiency - a lack of essential element(s) needed for plant life
rhizobia - soil bacteria that symbiotically interact with legume roots to form nodules and fix nitrogen
symbiosis - an interaction between two organisms that benefits them both
Essential Nutrients
Plants require only light, water, and about 20 elements (Figure 4.5.1) to support all their biochemical needs. These 20 elements are called essential nutrients. To be considered an essential element, three criteria are required: 1) a plant cannot complete its life cycle without the element; 2) no other element can perform the function of the element; and 3) the element is directly involved in plant nutrition.
Essential Elements for Plant Growth | |
Macronutrients | Micronutrients |
Carbon (C) | Iron (Fe) |
Hydrogen (H) | Manganese (Mn) |
Oxygen (O) | Boron (B) |
Nitrogen (N) | Molybdenum (Mo) |
Phosphorus (P) | Copper (Cu) |
Potassium (K) | Zinc (Zn) |
Calcium (Ca) | Chlorine (Cl) |
Magnesium (Mg) | Nickel (Ni) |
Sulfur (S) | Cobalt (Co) |
Sodium (Na) | |
Silicon (Si) | |
All of the required mineral elements can potentially limit growth. The limitation can come about both because that element is lacking from the soil or because, although the element is present, it is unavailable because of soil conditions. For instance, iron is frequently unavailable in basic soils even though it may be present in abundance. The problem is that under aerobic, basic conditions very little iron is present in a form that readily dissolves.
Somewhere on earth, there are soils that are deficient in all of the 14 mineral elements required by plants and deficiencies can develop even for elements like molybdenum that are needed in very small amounts. In the early 19th century Carl Sprengel developed an idea later championed by Justus van Liebig called the ‘Law of the Minimum:’ that plant growth will be limited not by nutrient availability generally but by whatever nutrient is in the shortest supply relative to how much is needed. For example, although additions of nitrogen often increase plant growth, if there isn’t enough molybdenum available such additions will not result in any growth enhancements. One can think of growing crops to be like baking a cake: if the cake recipe calls for five ingredients, making a cake can be limited by any of the five ingredients, and a lack of one is not made up for by excesses in others. This is a very straightforward idea that applies in many situations. But it runs counter to the common idea that response to factors will always be the constant: ‘if a little bit is good then a lot must be better’ is generally not the case!
While too little of the essential nutrients can limit growth, too many of the same elements (toxicities) can also retard growth. The most common toxicities are the result of saline soils that have high levels of K, Ca, Cl, SO4 and Na but unique soil conditions (waterlogging) can also bring about toxicities in iron and manganese in non-saline soils.
Access for free at https://openstax.org/books/biology-2e/pages/31-1-nutritional-requirements-of-plants
Major and Minor Nutrients
The essential elements can be divided into two groups: macronutrients and micronutrients. Nutrients required by plants in larger amounts are called macronutrient. About half of the essential elements are considered macronutrients: carbon, hydrogen, oxygen, nitrogen, phosphorus, potassium, calcium, magnesium, and sulfur.
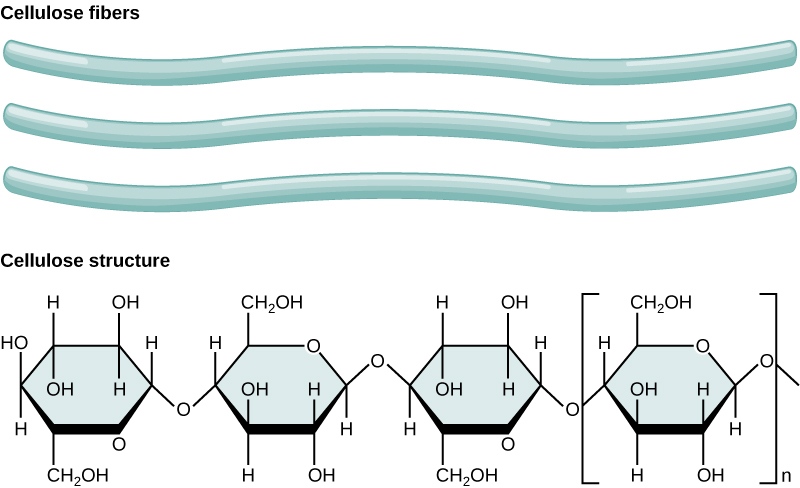
- The first of these macronutrients—carbon (C)—is required to form carbohydrates, proteins, nucleic acids, and many other compounds; it is therefore present in all macromolecules. On average, the dry weight (excluding water) of a cell is 50 percent carbon. Carbon is a key part of plant biomolecules. A biomolecule is any organic compound that is produced by living organisms. Figure 4.5.2 shows three cellulose fibers and the chemical structure of cellulose. Cellulose consists of unbranched chains of glucose subunits that form long, straight fibers.
The next most abundant element in plant cells is nitrogen (N); it is part of proteins and nucleic acids. Nitrogen is also used in the synthesis of some vitamins.
Hydrogen (H) and oxygen (O) are macronutrients that are part of many organic compounds, and together they form water. Oxygen is necessary for cellular respiration; plants use oxygen to store energy in the form of ATP.
Plants use their roots to uptake phosphorus, another macromolecule, from the soil as inorganic phosphate (Pi) in the forms of HPO42− or H2PO4− ions. Phosphorus (P) is necessary to synthesize nucleic acids and phospholipids. As part of ATP, phosphorus enables food energy to be converted into chemical energy through oxidative phosphorylation. Likewise, light energy is converted into chemical energy during photophosphorylation in photosynthesis, and into chemical energy to be extracted during respiration.
Sulfur (S) is part of certain amino acids, such as cysteine and methionine, and is present in several coenzymes. Sulfur also plays a role in photosynthesis as part of the electron transport chain, where hydrogen gradients play a key role in the conversion of light energy into ATP.
Potassium (K) is important because of its role in regulating stomatal opening and closing. As the openings for gas exchange, stomata help maintain a healthy water balance; a potassium ion pump supports this process.
Magnesium (Mg) and calcium (Ca) are also important macronutrients. The role of calcium is twofold: 1) to regulate nutrient transport, and 2) to support many enzyme functions.
Magnesium is important to the photosynthetic process. These minerals, along with the micronutrients, which are described below, also contribute to the plant’s ionic balance.
In addition to macronutrients, organisms require various elements in small amounts. These micronutrients, or trace elements, are present in very small quantities. The seven main micronutrients include boron, chlorine, manganese, iron, zinc, copper, and molybdenum. Most micronutrients are necessary for enzyme function. Nutrient deficiency, a lack of essential element(s) needed for plant life, can result in visible issues in plants.
- Boron (B) is believed to be involved in carbohydrate transport in plants; it also assists in metabolic regulation. Boron deficiency will often result in bud dieback.
- Chlorine (Cl) is necessary for osmosis and ionic balance; it also plays a role in photosynthesis. On some plant species, the most commonly described symptom of Cl deficiency is wilting of leaves, especially at the margins. As the deficiency progresses and becomes more severe, the leaves exhibit curling, bronzing, chlorosis, and necrosis.
- Copper (Cu) is a component of some enzymes. Symptoms of copper deficiency include browning of leaf tips and chlorosis (yellowing of the leaves).
- Iron (Fe) is essential for chlorophyll synthesis, which is why an iron deficiency results in chlorosis.
- Manganese (Mn) activates some important enzymes involved in chlorophyll formation. Manganese-deficient plants will develop chlorosis between the veins of its leaves. The availability of manganese is partially dependent on soil pH.
- Molybdenum (Mo) is essential to plant health as it is used by plants to reduce nitrates into usable forms. Some plants use molybdenum for nitrogen fixation; thus, it may need to be added to some soils before seeding legumes.
- Zinc (Zn) participates in chlorophyll formation and also activates many enzymes. Symptoms of zinc deficiency include chlorosis and stunted growth.
Macronutrients and micronutrients are both important for plant health. A lack of macronutrients and micronutrients will signal an issue with symptoms. Deficiencies in any of these nutrients—particularly the macronutrients—can adversely affect plant growth (Figure 4.5.3). Depending on the specific nutrient, a lack can cause stunted growth, slow growth, or chlorosis (yellowing of the leaves). Extreme deficiencies may result in leaves showing signs of cell death.
Explore nutrient deficiencies here or at this site.

Access for free at https://openstax.org/books/biology-2e/pages/31-1-nutritional-requirements-of-plants
Nitrogen Fixation
Nitrogen is an important macronutrient because it is part of nucleic acids and proteins. Atmospheric nitrogen, which is the diatomic molecule N2, or dinitrogen, is the largest pool of nitrogen in terrestrial ecosystems. However, plants cannot take advantage of this nitrogen because they do not have the necessary enzymes to convert it into biologically useful forms. However, nitrogen can be “fixed,” which means that it can be converted to ammonia (NH3) through biological, physical, or chemical processes. Biological nitrogen fixation (BNF) is the conversion of atmospheric nitrogen (N2) into ammonia (NH3), exclusively carried out by prokaryotes, such as soil bacteria or cyanobacteria. Biological processes contribute 65 percent of the nitrogen used in agriculture. The following equation represents the process:
N2+16 ATP + 8 e− + 8 H+ → 2NH3 + 16 ADP + 16 Pi + H2
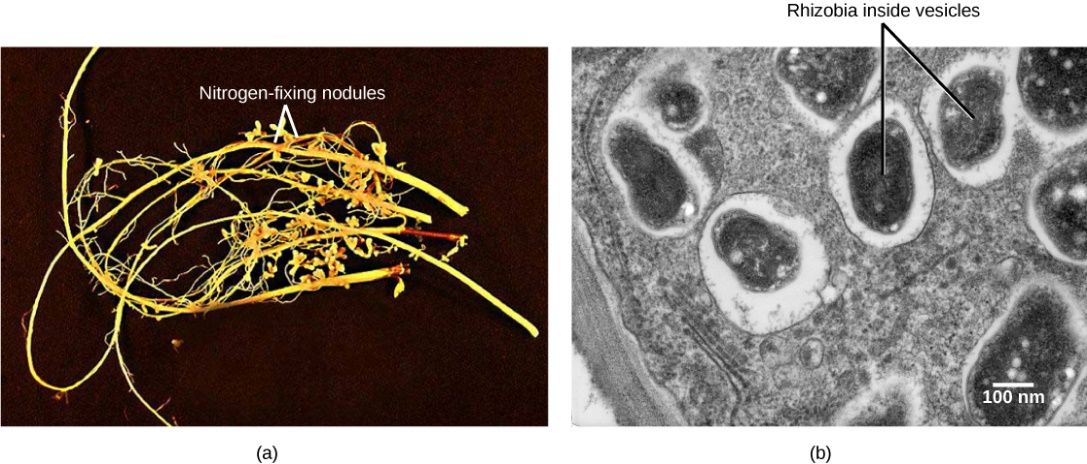
The most important source of BNF is the symbiotic interaction between soil bacteria and legume plants, including many crops important to humans (Figure 4.5.4). The NH3 resulting from fixation can be transported into plant tissue and incorporated into amino acids, which are then made into plant proteins. Some legume seeds, such as soybeans and peanuts, contain high levels of protein and serve among the most important agricultural sources of protein in the world.

Through symbiotic nitrogen fixation, the plant benefits from using an endless source of nitrogen from the atmosphere. The process simultaneously contributes to soil fertility because the plant root system leaves behind some of the biologically available nitrogen. Soil bacteria, collectively called rhizobia, symbiotically interact with legume roots to form specialized structures called nodules, in which nitrogen fixation takes place. This process entails the reduction of atmospheric nitrogen to ammonia, by means of the enzyme nitrogenase. Therefore, using rhizobia is a natural and environmentally friendly way to fertilize plants, as opposed to chemical fertilization that uses a nonrenewable resource, such as natural gas.. As in any symbiosis, both organisms benefit from the interaction: the plant obtains ammonia, and bacteria obtain carbon compounds generated through photosynthesis, as well as a protected niche in which to grow (Figure 4.5.5). Part a from Figure 4.5.5 is a photo of legume roots, which are long and thin with hair-like appendages. Nodules are bulbous protrusions extending from the root. Part B is a transmission electron micrograph of a nodule cell cross section. Black oval-shaped vesicles containing rhizobia are visible. The vesicles are surrounded by a white layer and are scattered unevenly throughout the cell, which is gray.
Access for free at https://openstax.org/books/biology-2e/pages/31-3-nutritional-adaptations-of-plants
Mycorrhizae: The Symbiotic Relationship between Fungi and Roots
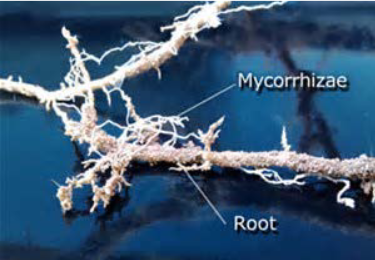
A nutrient depletion zone can develop when there is rapid soil solution uptake, low nutrient concentration, low diffusion rate, or low soil moisture. These conditions are very common; therefore, most plants rely on fungi to facilitate the uptake of minerals from the soil. Fungi form symbiotic associations called mycorrhizae with plant roots, in which the fungi actually are integrated into the physical structure of the root. The fungi colonize the living root-tissue during active plant growth.
Mycorrhizae functions as a physical barrier to pathogens. It also provides an induction of generalized host defense mechanisms, and sometimes involves production of antibiotic compounds by the fungi. Through mycorrhization, the plant obtains mainly phosphate and other minerals, such as zinc and copper, from the soil. The fungus obtains nutrients, such as sugars, from the plant root. Mycorrhizae help increase the surface area of the plant root system because hyphae, which are narrow, can spread beyond the nutrient depletion zone. Hyphae can grow into small soil pores that allow access to phosphorus that would otherwise be unavailable to the plant. The beneficial effect on the plant is best observed in poor soils. The benefit to fungi is that they can obtain up to 20 percent of the total carbon accessed by plants.
Access for free at https://openstax.org/books/biology-2e/pages/31-3-nutritional-adaptations-of-plants
Dig Deeper
Attributions
"Essential Nutrients for Plants" by Libretexts is licensed under CC BY-SA.
Inanimate Life by George M. Briggs is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License, except where otherwise noted.
"Mycorrhiza: The Hidden Plant Support Network" by the United States Department of Agriculture Natural Resources Conservation Service is in the Public Domain.
"Nutrition Needs and Adaptations" by Georgia Tech Biological Sciences is licensed under CC BY-NC-SA 3.0.
OpenStax Biology 2e by Mary Ann Clark, Matthew Douglas, and Jung Choi is licensed under CC BY 4.0.