
The video of Anion Exchange Chromatography created, explains the entire process in animated form with voiceover.
- Subject:
- Chemistry
- Material Type:
- Diagram/Illustration
- Author:
- Naba Patel
- Date Added:
- 10/04/2020

The video of Anion Exchange Chromatography created, explains the entire process in animated form with voiceover.
The purpose of this video lesson is to expand the student's knowledge about enzymes by introducing the antioxidant enzymes that are intimately involved in the prevention of cellular damage and eventual slowing of the aging process and prevention of several diseases. Students will learn that natural antioxidant enzymes are manufactured in the body and provide an important defense against free radicals. The topic of free radical action is introduced, covering how they are constantly generated in living cells both by ''accidents of chemistry'' and also by specific metabolic processes.

Practical lab into the chemical reaction between calcium carbide and water and the resulting ignition of acetylene gas.
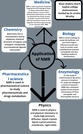
The diagram of NMR created, which explains the different application of NMR.

In this problem set students are given Rb/Sr and 87Sr/86Sr data for whole rock and mineral samples from three granitic intrusions in the Sierra Nevada. They use these data (in EXCEL) to calculate isochron ages and initial ages for the intrusions and then interpret their results. This problem is intended to teach some spreadsheet skills (linear regressions, graphing) as well as having them think about the use of radiogenic isotopes.
(Note: this resource was added to OER Commons as part of a batch upload of over 2,200 records. If you notice an issue with the quality of the metadata, please let us know by using the 'report' button and we will flag it for consideration.)
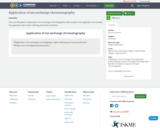
This is an illustration of Application of ion exchange Chromatography made using the miro application and includes the application that is water softining and protein purifiction.

It's an infograph vocalising the use of polymers in space based on verified research done.

This course details the quantitative treatment of chemical processes in aquatic systems such as lakes, oceans, rivers, estuaries, groundwaters, and wastewaters. It includes a brief review of chemical thermodynamics that is followed by discussion of acid-base, precipitation-dissolution, coordination, and reduction-oxidation reactions. Emphasis is on equilibrium calculations as a tool for understanding the variables that govern the chemical composition of aquatic systems and the fate of inorganic pollutants.
This course is offered through The MIT/WHOI Joint Program. The MIT/WHOI Joint Program is one of the premier marine science graduate programs in the world. It draws on the complementary strengths and approaches of two great institutions: the Massachusetts Institute of Technology (MIT) and the Woods Hole Oceanographic Institution (WHOI).
This video segment adapted from Building Big illustrates the strength of the arch in bridge design and construction.

This informational text introduces students to similarities and differences in the North and South Poles. The text is written at a grade one through two reading level. This is a PDF containing the informational text and a glossary.
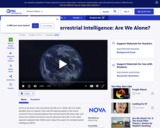
This video segment adapted from NOVA features a variety of scientific perspectives on the age old question, "Are we alone in the universe?" Animations make vivid the improbability that we could intercept a radio wave signaling extra terrestrial intelligence.

This resource is a video abstract of a research paper created by Research Square on behalf of its authors. It provides a synopsis that's easy to understand, and can be used to introduce the topics it covers to students, researchers, and the general public. The video's transcript is also provided in full, with a portion provided below for preview:
"It’s a feared moment for every scientist: the discovery that years of painstaking research has led to results that can't be repeated. Many think that poorly characterized antibodies have contributed to this reproducibility crisis more than any other laboratory tool. A new study published in Molecular Cell supports this hypothesis, at least in the context of chromatin immunoprecipitation. Although accurate ChIP interpretation depends on near-perfect antibody specificity, the report shows that many of these reagents are far less capable than their advertising suggests, which calls into question several widely accepted paradigms on genomic regulation. The study focused on histone post-translational modifications; specifically all three methylation states of lysine 4 on histone H3. Through ChIP experiments, H3K4 methylation has been strongly linked to transcriptional control..."
The rest of the transcript, along with a link to the research itself, is available on the resource itself.
This artcile is about Arsenic poisoning
This article and slide show from the New York Times, features several scientists from the University of Alaska, Fairbanks, who study the effects of thawing permafrost in Alaska.

In this laboratory exercise students will have an opportunity to examine the crystal structures, optical properties and health hazards of the common asbestos minerals. The laboratory will reinforce optical microscopic skills that students have learned in mineralogy and show them how mineralogy can be critical to understanding a current public policy issue.
(Note: this resource was added to OER Commons as part of a batch upload of over 2,200 records. If you notice an issue with the quality of the metadata, please let us know by using the 'report' button and we will flag it for consideration.)
In this interactive activity from ChemThink, take a closer look at atomic structure, properties, and behaviors.

Explore the interactions between various combinations of two atoms. Turn on the force arrows to see either the total force acting on the atoms or the individual attractive and repulsive forces. Try the "Adjustable Attraction" atom to see how changing the parameters affects the interaction.

Explore the interactions between various combinations of two atoms. Turn on the force arrows to see either the total force acting on the atoms or the individual attractive and repulsive forces. Try the "Adjustable Attraction" atom to see how changing the parameters affects the interaction.

In this hands-on OLogy activity, kids learn about matter by building their own models of carbon out of pipe cleaners, wire, and clay. The activity begins with a kid-friendly introduction to matter, elements, and atoms. The illustrated, step-by-step directions show how to use the information about carbon on the Periodic Table to create a mobile that shows the element's basic structure. A PDF version of the Periodic Table, along with a kid-friendly overview of how to read it, is also included.

Reviews middle school chemistry for getting information about atomic structure and periodic table grouping.